
Concept explainers
(a)
Interpretation:
The parent heterocyclic base in the given compound has to be determined.
Concept introduction:
Heterocyclic amine is a five or six membered cyclic organic compound which contains one or more nitrogen atoms.
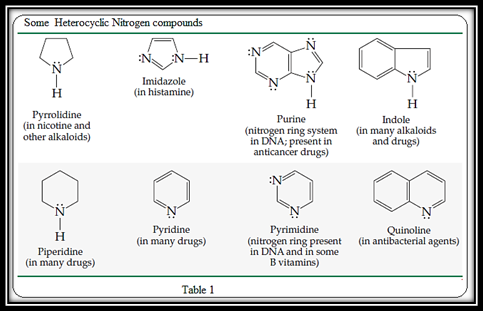
Figure 1
(b)
Interpretation:
The
Concept introduction:
Organic compounds are classified by the presence of characteristic functional groups.
A functional group is an atom or a group of atoms other than a
Functional groups have a specific characteristics, name, formula and structure.
Some functional groups are listed below,
Want to see the full answer?
Check out a sample textbook solution
Chapter 16 Solutions
Fundamentals of General, Organic, and Biological Chemistry (8th Edition)
- Salol, the phenyl ester of salicylic acid, is used as an intestinal antiseptic. Draw the structure of phenyl salicylate.arrow_forwardFor the first part, draw a Fischer projection formula for the enantiomer of each of the following monosaccharides. For the second part, identify whether the images are in D- or L-configuration. Write your answers first on a piece of bond paper.arrow_forwardWhy is the structure of ornithine so similar to the structure of citrulline?arrow_forward
- D- and L- designations are used to distinguish between the two possible enantiomers of the monosaccharide, galactose. From the Fischer projection, determine the designation of this monosaccharide.arrow_forwardWhich of the following define the stereochemistry of alanine (as per the structure shown)? Note: Functional groups arranged horizontally are facing towards the front, and the functional groups arranged vertically are facing towards the back. a) d- b) S- c) R- d) I-arrow_forwardWhy is this compound achiral?arrow_forward
- (a) (S)-2-chlorobutane, draw a three-dimensional representation.arrow_forwardDraw the four stereoisomers of threonine as Fischer projections.arrow_forward(A) What are Waxes? Draw the structure of wax, which is made up of palmitic acid (16:0) and a saturated 18-carbon alcohol. (В) Consider the structure of menthol. How many isoprene units are present in menthol? (C) What type of isoprene linkage (head-to-tail or tail-to-tail) is present in menthol? Identify the isoprene linkage in the following structure of menthol and indicate by a circle. Menthol =arrow_forward
- Draw the structure of the tripeptide alanylglycylvaline and determine its name using three-letter abbreviations.arrow_forwardWhat is unusual about glutathione’s structure? (If you cannot answer this question, draw the structure you would expect for the tripeptide and compare your structure with the actual structure.)arrow_forwardGlutathione, a powerful antioxidant that destroys harmful oxidizing agents in cells, is composed of glutamic acid, cysteine, and glycine, and has the following structure. a.) What product is formed when glutathione reacts with an oxidizing agent?b.) What is unusual about the peptide bond between glutamic acid and cysteine?arrow_forward
 BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman
BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman
Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY
Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON
Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON





