
(a)
Interpretation:
The given amine has to be named and it should be indicated whether as the ammonium salt of a primary, secondary or tertiary amine.
Concept introduction:
In chemistry Structure is the arrangement of
Depending on the number of carbon side chain of the nitrogen, different types of amines can form.
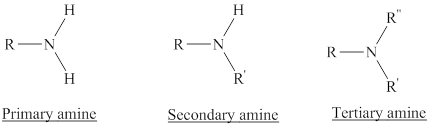
From the name of the compound its structure can be determined.
Primary amines can be named in the IUPAC system in several ways,
For simple amines the suffix – amine is added to the name of the alkyl substituent.
The suffix-amine can be used in place of the final –e in the name of the parent compound.
For a secondary amine an N prefixes the compound giving the shorter carbon chain and its chain prefix name.
For a tertiary amine an N, N prefixes the compound giving the two shorter carbon chains and their side chain prefix names.
Ammonia is very soluble in water giving an alkaline solution which can be neutralized by acids to produce ammonium salts.
(b)
Interpretation:
The structure of the given compound has to be drawn and it should be indicated whether as the ammonium salt of a primary, secondary or tertiary amine.
Concept introduction:
In chemistry Structure is the arrangement of chemical bonds between atoms in a molecule, specifically which atoms are chemically bonded to what other atoms with what kind of chemical bond.
Amines are the derivatives of ammonia
Depending on the number of carbon side chain of the nitrogen, different types of amines can form.

From the name of the compound its structure can be determined.
Primary amines can be named in the IUPAC system in several ways,
For simple amines the suffix – amine is added to the name of the alkyl substituent.
The suffix-amine can be used in place of the final –e in the name of the parent compound.
For a secondary amine an N prefixes the compound giving the shorter carbon chain and its chain prefix name.
For a tertiary amine an N, N prefixes the compound giving the two shorter carbon chains and their side chain prefix names.
Ammonia is very soluble in water giving an alkaline solution which can be neutralized by acids to produce ammonium salts.
Heterocyclic amine is a five or six membered cyclic organic compound which contains one or more nitrogen atoms.
(c)
Interpretation:
The structure of the given compound has to be drawn and it should be indicated whether as the ammonium salt of a primary, secondary or tertiary amine.
Concept introduction:
In chemistry Structure is the arrangement of chemical bonds between atoms in a molecule, specifically which atoms are chemically bonded to what other atoms with what kind of chemical bond.
Amines are the derivatives of ammonia
Depending on the number of carbon side chain of the nitrogen, different types of amines can form.

From the name of the compound its structure can be determined.
Primary amines can be named in the IUPAC system in several ways,
For simple amines the suffix – amine is added to the name of the alkyl substituent.
The suffix-amine can be used in place of the final –e in the name of the parent compound.
For a secondary amine an N prefixes the compound giving the shorter carbon chain and its chain prefix name.
For a tertiary amine an N, N prefixes the compound giving the two shorter carbon chains and their side chain prefix names.
Ammonia is very soluble in water giving an alkaline solution which can be neutralized by acids to produce ammonium salts.
Want to see the full answer?
Check out a sample textbook solution
Chapter 16 Solutions
Fundamentals of General, Organic, and Biological Chemistry (8th Edition)
- Propose a scheme to separate and isolate a mixture containing 4-aminobenzoate, 1,2,4,5-tetrachlorobenzene, and napthalene. Isolate two of the three solids by extraction. (please explain what solvents to use and the process step by step)arrow_forwardDraw structural formulas for all of the following. Q.) Alcohols with the molecular formula C4H10Oarrow_forwardIdentify the functional groups in the following compoundsarrow_forward
- Draw a Fischer projection formula for the enantiomer of each of the following monosaccharides. (a to d)arrow_forwardA decapeptide has the following acid composition; Al2,Arg,Cys,Glu,Leu,Phe,Val Partial hydrolysis yield the following tripeptides; Cys-Glu-Leu+Gly-Arg-Cys+Leu-Ala-Ala+Lys-Val-Phe-Gly Reaction of the decapeptide with 2,4-dinitrofluorobenzene yields 2,4-dinitrophenylysine. From the experimental data, deduce the primary structure of the decapeptide. Suggest a scheme you will follow to synthesize the dipeptide Ala-Gly.arrow_forwardWrite the formulas of the four singly chlorinated isomers formed when 2-methylbutane reacts with Cl2 in the presence of light.arrow_forward
- what is the crystallinity of phenazopyridine? with illustationarrow_forwardMethadone is dispensed as its hydrochloride salt. What is the use of the salt rather than the amine?arrow_forwardDrawn are four isomeric dimethylcyclopropanes. How are the compounds in each pair related (enantiomers, diastereomers, constitutional isomers): A and B; A and C; B and C; C and D?arrow_forward
 BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman
BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman
Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY
Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON
Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON





