
Concept explainers
Interpretation:
It should be determined that whether the given substance reacts with aqueous hydrochloric acid or not. If it reacts with
Concept introduction:
Depending on the number of carbon side chain of the nitrogen, different types of amines can form.
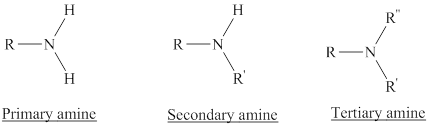
Reaction of amines and acid will give amine salt (ammonium ion).
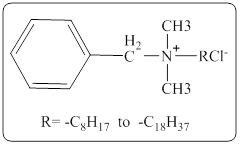
In a quaternary ammonium ion a nitrogen atom with four attached groups is positively charged. And their compounds are known as quaternary ammonium salt.
One commonly encountered quaternary ammonium salt has the following structure,
Want to see the full answer?
Check out a sample textbook solution
Chapter 16 Solutions
Fundamentals of General, Organic, and Biological Chemistry (8th Edition)
- Using the picture provided, match the correct name of each of the functional groups highlighted in blue.arrow_forwardFor the first part, draw a Fischer projection formula for the enantiomer of each of the following monosaccharides. For the second part, identify whether the images are in D- or L-configuration. Write your answers first on a piece of bond paper.arrow_forwardColumn A shows the names of some of the c functional groups. Column B shows their structure. Match each entry in column A one in column B.arrow_forward
- IDENTIFY THE FUNCTIONAL GROUP PRESENT IN THESE COMPOUNDS. A = ? B = ? C = ?arrow_forwardAssuming equal concentrations of conjugate base and acid, which one of the following mixtures is suitable for making a buffer solution with an optimum pH of 7.4–7.6? NaOCl / HOCl (K a = 3.2 × 10 –8) NH 3 / NH 4Cl (K a = 5.6 × 10 –10) NaNO 2 / HNO 2 (K a = 4.5 × 10 –4) NaCl / HCl CH 3COONa / CH 3COOH (K a = 1.8 × 10 –5)arrow_forwardOne of these forms of cocaine is relatively insoluble inwater: which form, the free base or the hydrochloride?arrow_forward
- Drawn are four isomeric dimethylcyclopropanes. How are the compounds in each pair related (enantiomers, diastereomers, constitutional isomers): A and B; A and C; B and C; C and D?arrow_forwardIn the formula for sucrose (structure shown above), the bottom ring is pyranose. True or False?arrow_forwardThe liquids butan-1-ol and butanal have similar molar masses. Which is expected to have the higher boiling point? Explain your choices.arrow_forward
- Give the equation for the complete titration of aspartic acid with a base, NaOH. At what pH, can you use aspartate solution a buffer?arrow_forwardFumaric acid is a metabolic intermediate that has the systematic name trans-2-butenedioic acid. Draw its structure.arrow_forwardIdentify the acid on the left and its conjugate base on the right in the following equations:(a) HOCl + H2O ↔ H3O+ + OCl-(b) HONH2 + H2O ↔ HONH3+ + OH-(c) NH4+ + H2O ↔ NH3 + H3O+(d) 2HCO3-2 ↔ H2CO3 + CO3-2 (e) PO4-3 + H2PO4- ↔ 2HPO4-2arrow_forward
 BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman
BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman
Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY
Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON
Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON





