
(a)
Interpretation:
The given reaction equation should be completed.
Concept introduction:
Depending on the number of carbon side chain of the nitrogen, different types of amines can form.
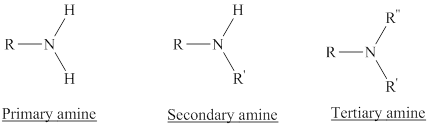
Reaction of amines and acid will give amine salt an (ammonium ion).
(b)
Interpretation:
The given reaction equation should be completed.
Concept introduction:
Amines are the derivatives of ammonia
Depending on the number of carbon side chain of the nitrogen, different types of amines can form.

Reaction of amines and acid will give amine salt an (ammonium ion).
Amine are weak bases (proton
(c)
Interpretation:
The given reaction equation should be completed.
Concept introduction:
Amines are the derivatives of ammonia
Depending on the number of carbon side chain of the nitrogen, different types of amines can form.

Reaction of amines and acid will give amine salt an (ammonium ion).
Treating an amine salt with a strong base regenerates the “parent” amine.
Want to see the full answer?
Check out a sample textbook solution
Chapter 16 Solutions
Fundamentals of General, Organic, and Biological Chemistry (8th Edition)
- Using condensed structures, supply an outline of the mechanism for the formation of Gly-Gly in the human body. (Hint: It is not a simple condensation reaction).arrow_forwardPredict the missing reactant of this biochemical reaction: X + Pi P-O + H₂O That is, in the drawing area below, draw the chemical structure of the reactant molecule X. Note for advanced students: you can assume any necessary small-molecule reactants, like water, are available. Click and drag to start drawing a structure. X :0 Śarrow_forwardBiotin (below), also known as Vitamin B7, is a very large organic compound derived primarily from meats, eggs, nuts, and certain vegetables. The pKa of biotin is 4.4. This means that biotin is/is not soluble at the pH of stomach acid (pH ~2.5) and is/is not soluble at the pH of duodenum (pH ~6.1). a is soluble in stomach and duodenum b is soluble in stomach but not duodenum c is not soluble in either stomach or duodenum d is not soluble in stomach but is soluble in duodenumarrow_forward
- Make use of the table below in answering the questions asked: Amino acid pK₁ pK₂ pK, Isoleucine 2.32 9.76 Leucine 2.32 9.74 Lysine 2.16 9.06 10.54 Tyrosine 2.20 9.21 10.46 1. Kindly draw manually the structure of tripeptic ILY at pH 7.00 2. Kindly draw manually the most protonated structure of tripeptide ILYarrow_forwardSerine is an uncharged, polar amino acid (below and left). Threonine (below and right) is another uncharged, polar amino acid. Illustrate the hydrolysis of a dipeptide that was made from these two amino acids. Be sure to write all involved reactants and products in the structural form like Serine, and as a proper chemical equation; eg. reactant(s) – product(s). Your diagram of the dipeptide must show the N-terminal and C-terminal ends at opposite sides of the dimer molecule, and you need to indicate where the peptide bond is with an arrow/highlighter. 2. H C-N OH NH, но CH, CH,-CH -C-COOH онarrow_forwardConsider olive oil, an oil with a high percentage of fat derived from oleic acid (otherwise known as cis[18:1] fatty acid). a, Explain why such a structure may allow olive oil to be one of the "healthier" oils? b, why such a structure may allow olive oil to be a liquid at room temperature, while butter and lard are solid at room temperature? c, why such a structure may cause olive oil to be prone to oxidative damage upon exposure to air and heat?arrow_forward
- Consider a protein in which a negatively charged glutamic acid side chain (pKa=4.2) makes a salt bridge (ion-ion interaction) with a positively charged histidine side chain (pKa=6.5). Part C: Justify your answer with calculations of partial charges on these amino acid side chains at pH=7.9.(Hint: Consider lessons from Coulomb's law, and the Henderson-Hasselbalch equation.)arrow_forwardThe following amino acid is dissolved in solution at pH 7.4. HC H' + CH CH₂ H/ H+ HC a. Which amino acid is this? (all lower case) N- H CH CH₂ b. What is the pKa of its ionizable side chain? (Use the value from the textbook.) c. What is the ratio of the uncharged to the charged form at pH 7.4? (write answer as number with one decimal place, ex 2.5 or 0.2 or 50.0 with an implicit "to 1")arrow_forwardUsing the picture provided, match the correct name of each of the functional groups highlighted in blue.arrow_forward
- Give one specific sample structure (Fischer Projection or Haworth) of the following carbohydrate derivatives: - Amino-sugararrow_forwardDraw a diagram of the histidine side chain in the protonated and unprotonated states. Show the contributing resonance structures and the composite resonance structure for the protonated state.arrow_forwardWhich of the following define the stereochemistry of alanine (as per the structure shown)? Note: Functional groups arranged horizontally are facing towards the front, and the functional groups arranged vertically are facing towards the back. СООН + H₂N to OS- Od- CH, OR-arrow_forward
 BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman
BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman
Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY
Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON
Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON





