
a.
Interpretation:
Amide given below has to be named.
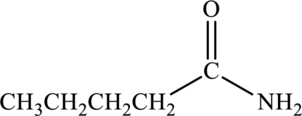
Concept Introduction:
Amides contain a nitrogen atom that is bonded to carbonyl group. The nitrogen atom that is bonded to the carbonyl group may be bonded to alkyl groups or hydrogen atoms. Depending on the number of carbon atoms that is attached to the nitrogen atom, the amide is classified as primary, secondary, and tertiary.
Naming of amides are done using specific rules:
Primary amides are named by considering the parent
Secondary and tertiary amide contains two parts in its name. The alkyl groups that are bonded to the nitrogen atom is named using N- as the prefix for the alkyl group and this precedes the name. The
b.
Interpretation:
Amide given below has to be named.
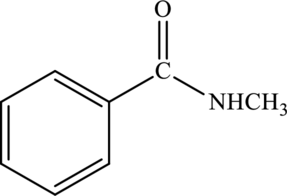
Concept Introduction:
Refer part “a.”.
c.
Interpretation:
Amide given below has to be named.
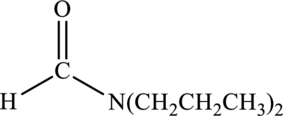
Concept Introduction:
Refer part “a.”.
Want to see the full answer?
Check out a sample textbook solution
Chapter 13 Solutions
Principles of General, Organic, Biological Chemistry
- Name each amine. CH;-CH,-N-CH,-CH, b. CH3-CH,-CH2-N-CH3 H CH3 CH3-CH2-CH2-N-CH2-CH;-CH2-CH3arrow_forwardTrue or False Amine salts have increased solubility in water. Amines have a pleasant floral aroma. Carboxylic acids have significantly higher boiling points than alcohols of comparable molecular weight Butanal would have a higher boiling point than butanoic acid. Pentanone would have a lower boiling point than pentane. Carboxylate ions have decreased solubility in water.arrow_forwardWhat is an acetal?arrow_forward
- amines Name and classify each compoundarrow_forwardWhat reactants will product this amide? Draw the reactionarrow_forwardN-p-hydroxyphenylethanamide is commonly known as a. acetaminophen b. acetamide c. acetanilide d. formamide High molar mass amines have __________ odor. a.strong ammoniacal b.fruity c.fishy d.obnoxious Trimethyl amine has _________ odor. a.obnoxious b.fishy c. ammoniacal d. fruityarrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
