
Concept explainers
a.
Interpretation:
Compound that has higher boiling point in the below pair has to be identified.
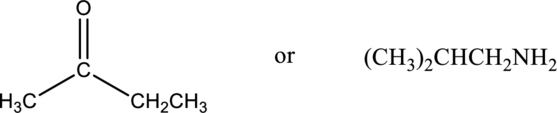
Concept Introduction:
Boiling point of a substance is the temperature at which the liquid phase gets converted into vapor phase. Intermolecular forces plays an important role in the boiling point of a substance. If the intermolecular force is stronger, then the boiling point will be higher. Boiling point of compound that can form hydrogen bonding and polar will be greater than that of the compounds that cannot form hydrogen bonding but can be polar. Boiling point of polar compounds are higher than that of the nonpolar compounds.
b.
Interpretation:
Compound that has higher boiling point in the below pair has to be identified.
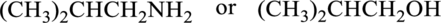
Concept Introduction:
Refer part “a.”.
b.
Interpretation:
Compound that has higher boiling point in the below pair has to be identified.
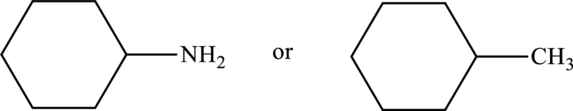
Concept Introduction:
Refer part “a.”.
Want to see the full answer?
Check out a sample textbook solution
Chapter 13 Solutions
Principles of General, Organic, Biological Chemistry
- Draw the products formed when phenol(C6H5OH) is treated with each reagent. Give an explanation. d. (CH3CH2)2CHCOCl, AlCl3 j. product in (d), then NH2NH2, – OHarrow_forwardGive the IUPAC name for each compound. OH a. CH3CH(CH₂)4CH3 (select) OH (CH3CH₂)2CHCHCH₂CH3 (select) b. C. d. CH3 OH (select) (select) (select) OH (select) (select) (select)arrow_forwardRank the following compounds in order of increasing boiling point.arrow_forward
- 1. What letter has an aldehyde with molecular formula C₄H₈O. 2. What letter has an ester with molecular formula C₄H₈O₂ 3. What letter has a ketone with molecular formula C₄H₈O₂ 4. What letter has a carboxylic acid with molecular formula C₄H₈O₂arrow_forwardWhich compound in each pair has the higher boiling point?arrow_forwardArrange the following compounds in order of increasing boiling point.Explain. (CH3)2CHCH2OH (CH3)2CH(CH2)2CH2OH (CH3)2CH(CH2)3CH2OHarrow_forward
- Draw the products formed when phenol(C6H5OH) is treated with each reagent. Give an explanation. c. CH3CH2Cl, AlCl3 l. product in (c), then KMnO4arrow_forwardDiethyl ether (CH3CH2OCH2CH3) and butan-1-ol (CH3CH2CH2CH2OH) are constitutional isomers with the same molecular formula, however their boiling points are very different. Which has the higher boiling point? O Diethyl Ether O Butan-1-olarrow_forwardDraw the products formed when phenol (C6H5OH) is treated with each set of reagents. a. [1] HNO3, H2SO4; [2] Sn, HCl b. [1] (CH3CH2)2CHCOCl, AlCl3; [2] Zn(Hg), HCl c. [1] CH3CH2Cl, AlCl3; [2] Br2, hν d. [1] (CH3)2CHCl, AlCl3; [2] KMnO4arrow_forward
- Which alcohol is the most soluble in water? A CH;CH,CHOHCH3 B CH;CH2CH2CH2OH (c) (CH3)2CHCH2OH D (CH3)3COHarrow_forwardWhy is the boiling point of propane-1,3-diol (HOCH2CH2CH2OH) higher than the boiling point of propane-1,2-diol [HOCH2CH(OH)CH3] (215 °C vs. 187 °C)? Why do both diols have a higher boiling point than butan-1-ol (CH3CH2CH2CH2OH, 118 °C)?arrow_forwardTHC is the active component in marijuana, and ethanol is the alcohol in alcoholic beverages. Explain why drug screenings are able to detect the presence of THC but not ethanol weeks after these substances have been introduced into the body. CH3 OH CH;CH2-OH ethanol CH3 ČH3 (CH),CH3 tetrahydrocannabinol THCarrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
