
Concept explainers
a.
Interpretation:
The ester that is formed when the given
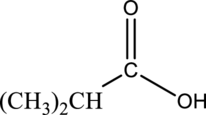
Concept Introduction:
In presence of acid, the carboxylic acid and an alcohol reacts to form ester as product. Acid acts as a catalyst in this reaction. This is known as Fischer esterification reaction. Esterification reaction is an example of substitution reaction because, the
b.
Interpretation:
The ester that is formed when the given carboxylic acid reacts with ethanol in presence of sulfuric acid has to be drawn.
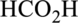
Concept Introduction:
Refer part “a.”.
c.
Interpretation:
The ester that is formed when the given carboxylic acid reacts with ethanol in presence of sulfuric acid has to be drawn.
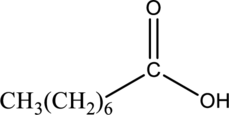
Concept Introduction:
Refer part “a.”.
d.
Interpretation:
The ester that is formed when the given carboxylic acid reacts with ethanol in presence of sulfuric acid has to be drawn.
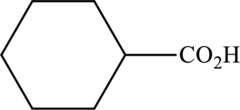
Concept Introduction:
Refer part “a.”.
Want to see the full answer?
Check out a sample textbook solution
Chapter 13 Solutions
Principles of General, Organic, Biological Chemistry
- H3C. H3C CH3 Br 1-propanolarrow_forward3) Which aa has the strongest acid side chain? a) b) H2N -CH-ö OH H,N -CH-C- OH CH2 ČH2 CH2 CH2 OH NH2 c) d) H2N -CH-ö OH H2N- CH OH CH2 CH-OH ČH3 OH 4) In order to oxidize an alcohol to aldehyde we use А) РСС В) НCL С) НЗО+arrow_forwardGive the IUPAC name for each ketone. C-CHCH,CH3 b. a.arrow_forward
- 8 What ester is formed when each carboxylic acid is treated with 2-propanol [(CH 3) 2CHOH] in the presence of H 2SO 4?arrow_forwardCH,CH,CH,-OH Draw the structure of the product expected when each of the following alcohols is reacted with a sulfuric acid catalyst au the temperature indicated. H,SO, CH,-CH-CH, 180°C a. OH H,SO, CH, — CH—СH,—ОН 180°C b. CH; H,SO, CH-CH-OH 140°C с. CH, CH,-CH-CH,-CH, H,SO, d. 140°C d. OHarrow_forwardDraw the products formed when phenol(C6H5OH) is treated with each reagent. Give an explanation. d. (CH3CH2)2CHCOCl, AlCl3 j. product in (d), then NH2NH2, – OHarrow_forward
- What is the reduction product of the following compound with H2/Pd? A 2-propyl-1-cyclohexanol B 2-propenyl-1-cyclohexanol C2-propylcyclohexanone D 1-propyl-2-cyclohexanol What is the oxidation product of this compound? H' A ethanoic acid acetic acid propanoic acid D propanone What is the oxidation product of this compound? A 3,4-dimethylpentanoic acid B 2,3-dimethylpentanoic acid C2,3-dimethylpentanone 3,4-dimethylpentanonearrow_forwardDraw the carbonyl products formed when each alcohol is oxidized with K 2Cr 2O 7.arrow_forward1. Draw the product formed when phenylacetaldehyde (C6H5CH2CHO) is treated with each reagent:arrow_forward
- 19) What is the IUPAC name for this compound? CH3 H3C- -сн—сH,—CH2--с— он O 2-methyl-4-butanoic acid pentanoic acid 2-methyl butyric acid 3-methylbutanoic acid O 2-methylbutanoic acidarrow_forwardDraw the skeletal structure of the ester formed when carboxylic acid CH3CH2CH2CH2CO₂H is treated with ethanol (CH3CH₂OH) in the presence of H2SO4 Click and drag to start drawing a structure. Garrow_forward7 8 11 12 me CH3 H3C 3-butylpropanoate 2-butoxypropane propyl-2-butanoate 2-butylpropanoate H₂ C CH3arrow_forward
 Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning

