
Concept explainers
a.
Interpretation:
The compound
Concept Introduction:
Ammonium salts are similar to ionic compounds. They have cation and anion. Ammonium salts are highly soluble in water.
b.
Interpretation:
The compound
Concept Introduction:
Refer part “a.”.
c.
Interpretation:
The compound
Concept Introduction:
Refer part “a.”.
d.
Interpretation:
The given compound is water soluble or water insoluble has to be labelled.
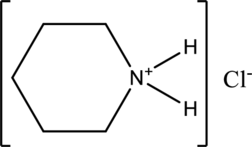
Concept Introduction:
Refer part “a.”.
Want to see the full answer?
Check out a sample textbook solution
Chapter 13 Solutions
Principles of General, Organic, Biological Chemistry
- Draw the possible products of this epoxide ring-opening reaction. Use a dash or wedge bond to indicate the stereochemistry of substituents on asymmetric centers, where applicableIgnore any inorganic byproducts. 41100 H 1) NaOH / H₂O 2) dilute HCI ☑Ⓒ Please select a drawing or reagent from the question areaarrow_forwardCurved arrows are used to illustrate the flow of electrons. Using the provided starting and product structures, draw the curved electron-pushing arrows for the following reaction or mechanistic steps. Be sure to account for all bond-breaking and bond-making steps. I I I I Mg. :O: Br. :O: Select to Add Arrows :O: CO2 THE H Mg 0: Br Please select a drawing or reagent from the question areaarrow_forwardWhich is the more stable base? a. Br− or I− b. CH3O− or CH3S− c. CH3CH2O− or CH3COO− d. H2C CH or HC C− e. FCH2CH2COO− or CH2CH2COO- f. CLCH2CH2O- or CL2CHCH2O-arrow_forward
- Draw the products of each acid-base reaction. H CH3 H CH,CH,NHCH3 COOH + а. NAOH b. CF3 + HCI CH,O naproxen anti-inflammatory agent fluoxetine antidepressant 1 1arrow_forwardShow how to bring about each conversion in good yield. a. b. C6H5 Cl OH COOH C6H5 COOHarrow_forwarda) Rank the following in order of increasing boiling point. HO CH3 A B b) Rank the following in order of increasing acidity. H3C `OH HS. но. `OH HO Aarrow_forward
- 4. Which of the following is the strongest base? A. (CH3)3COH B. CH3CH₂NH™ C. H₂O D. CH3NH2arrow_forwardDraw the products of each acid-base reaction. NaOH b. + NazCO3 a. + CH3CH,CH, HO, OHarrow_forwardRank the below compounds in order of increased acidity and explain your choice. HO. H3C || HO. HO.arrow_forward
- What is the structure of compound A? 010 Compound A tot 01 Oll O III O IV OV || s ||| Compound A IV COOHarrow_forward1. Rank each set of three alcohols in terms of increasing acidity, "1" being least acidic, and "3" being most acidic. goos CCI3 CI3C. Cl3 а. ОН ОН OH b. OH ОН ОН OH С. HOarrow_forwardOH 15. CH3-C-H ÓCH, The compound above is an example of a(n) a. acetal b. ketal O c. hemiacetal d. hemiketal e. none of the abovearrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
