
Concept explainers
(a)
Interpretation:
Structure of the molecule D should be converted to a skeletal structure.
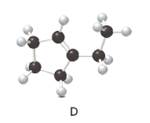 = (
= (
Concept Introduction:
Cyclic
(b)
Interpretation:
The resulting product should be identified after reacting
Concept Introduction:
Cyclic alkenes are hydrocarbon molecules that consist a carbon-carbon double bond which has the general formula of
Reaction of alkene with
Hydro halogenation reaction of alkenes follows the Markovnikov's rule.
(c)
Interpretation:
The resulting product should be identified after reacting
Concept Introduction:
Cyclic alkenes are hydrocarbon molecules that consist a carbon-carbon double bond which has the general formula of
Reaction of alkene with
Hydration reaction of alkenes follows the Markovnikov's rule.
Want to see the full answer?
Check out a sample textbook solution
Chapter 13 Solutions
General, Organic, and Biological Chemistry - 4th edition
- Draw the structures of the chief product formed when the following alcohols are dehydrated to alkenes: a. b.arrow_forwardName and draw a structural formula for the product of each alkene addition reaction. (a) (b)arrow_forwardDraw out the structures (condensed structural formulas) for the following reactions (reactants and products) and name the product formed. a) Hydrohalogenation of 1,3-dimethylcyclopentene with HBr Name product: b) Hydrogenation of 3,4-dimethylcyclobutene Name product: c) Hydration of 3-ethyl-2-pentene cturslomeo Name product: d) Alkylation of benzene with 2-chloropropane (isopropylchloride) Name product: e) Mono-halogenation of 2,2-dimethylbutane with Cl2 Name product:arrow_forward
- What organic compound can be produced from the oxidation of alcohols and aldehydes? alcohols carboxylic acids ethers esters What organic reaction is displayed in the picture below? * H H NaOH H C- H Ethanol heat H H Cl H- -H Alkyl halide substitution elimination oxidation addition alkenearrow_forwardDraw the structure of the alkene needed to synthesize the product shown using the reagents provided. Click and drag to start drawing a structure. 1. CH3CO₂H 2. NaOH, H₂O HO OHarrow_forwardExplain the Hydroboration–oxidation two-step reaction sequence that converts an alkene to an alcohol.arrow_forward
- 4. What is the major organic product obtained from the following reaction? 1. NaNH, 2. CH₂CH₂Brarrow_forwardDraw the products of combustion of each alkanearrow_forwardA hydrocarbon (Y) partially hydrogenates with the Lindlar's catalyst to form an alkene. When (Y) undergoes oxidative cleavage with ozone, it yields a single carboxylic acid product. The hydrocarbon (Y) is: 2-hexyne B) 2-hexen-1-ol 2-butyne D cyclohexanearrow_forward
- In general, which of the following statements correctly describes the reactivity of alkenes? Select one: A. An alkene is an electron-rich molecule and therefore can react as a nucleophile. B. Alkenes are generally unreactive compared alkanes. C. Alkenes react as electrophiles, whereas alkanes react as nucleophiles. D. Unlike alkynes, alkenes fail to undergo electrophilic addition reactions. Which of the following statements on bond-breaking (cleaving) or bond-forming is correct? Select one: A. In homolytic bond breaking, a bond is broken respectively by the movement of a pair of electrons in the same direction to one of the products. B. In heterolytic bond-forming, electrons flow from a nucleophile to an electrophile. C. Electron flow in homolytic bond breaking is shown by a double-headed arrow from the bond to a nucleophile. D. A double-headed arrow indicates the movement of an electron pair during homolytic bond breaking.arrow_forwardThe following reaction is an example of a reaction. нн H H Heat or light H-C-c-H + Cl-ci н—с—с—сі: + н—сi: нн нн ethane chloroethane O Substitution Elimination O Hydrogenation O Chlorinationarrow_forward1. Are addition reactions of alkenes exothermic or endothermic? 2. Do elimination reactions requiere a base or a nucleophile?arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

