
Concept explainers
(a)
Interpretation:
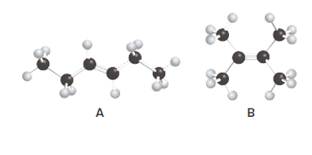
Structures of A and B should be identified as constitutional isomers, stereo isomers or not isomers of each other.
Concept Introduction:
Isomers are the different molecules that have the same molecular formula but differ in molecular arrangement in the space. There are several groups of isomers.
Stereoisomers are the form of isomers which have different three-dimensional orientations in the space.
Constitutional isomers are the form of isomers which have atoms bonded to each other in different ways.
(b)
Interpretation:
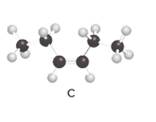
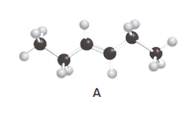
Structures of A and C should be identified as constitutional isomers, stereo isomers or not isomers of each other.
Concept Introduction:
Alkenes are hydrocarbon molecules that consist of a carbon-carbon double bond which has the general formula of
Isomers are the different molecules that have the same molecular formula but differ in molecular arrangement in the space. There are several groups of isomers.
Stereoisomers are the form of isomers which have different three-dimensional orientations in the space.
Constitutional isomers are the form of isomers which have atoms bonded to each other in different ways.
(c)
Interpretation:
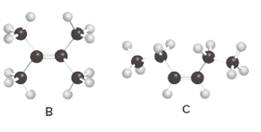
Structures of B and C should be identified as constitutional isomers, stereo isomers or not isomers of each other.
Concept Introduction:
Alkenes are hydrocarbon molecules that consist a carbon-carbon double bond which has the general formula of
Isomers are the different molecules which have the same molecular formula but differ in molecular arrangement in the space. There are several groups of isomers.
Stereoisomers are the form of isomers which have different three-dimensional orientations in the space.
Constitutional isomers are the form of isomers which have atoms bonded to each other in different ways.
Want to see the full answer?
Check out a sample textbook solution
Chapter 13 Solutions
General, Organic, and Biological Chemistry - 4th edition
- How are the following two compounds related to each other? H H3C KD H3C CH3 a) They are constitutional isomers of each other. O b) O c) H CH3 d) They are enantiomers of each other. They are stereoisomers of each other. They are functional isomers of each other.arrow_forwardClassify each pair of compounds as constitutional isomers, stereoisomers, identical molecules, or not isomers of each attached otherarrow_forwardLabel each pair as constitutional isomers, stereoisomers, or not isomers of each.arrow_forward
- Classify each pair of compounds as either identical, constitutional isomers, stereoisomers, or not isomers.arrow_forwardLabel each pair of compounds as constitutional isomers, stereoisomers, or not isomers of each other.arrow_forwardA Newman projection of a disubstituted cyclohexane is shown below. Determine which of the following 2D representations matches the Newman projection. H Br H H2 CH3 `H H H2 Br II Br CH3 \CH3 Brill. Br Br IV H3 II CH3 Br Br Br A) I B) II C) II D) IVarrow_forward
- Label attached pair of compounds as constitutional isomers, stereoisomers,or not isomers of each other.arrow_forwardLabel each pair of compounds with the appropriate label of their relationship: conformational isomers; stereoisomers, constitutional isomers, or two different compounds.arrow_forwardDraw the five constitutional isomers having molecular formula C6H14.arrow_forward
- Constitutional isomers or stereoisomers? b) CH3 CH3 «CH3 CH3 d) CIarrow_forwardDraw the ve constitutional isomers having molecular formula C6H14.arrow_forward12. Isomerism: Classify each pair of compounds below as either identical molecules, or constitutional isomers, or stereoisomers, or not isomers of each other. Place one of the following letter designations on the line beside each pair: I = identical C = constitutional isomers a. b. C. R R R S = stereoisomers N = not isomersarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





