
General, Organic, and Biological Chemistry - 4th edition
4th Edition
ISBN: 9781259883989
Author: by Janice Smith
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Question
Chapter 13, Problem 88P
Interpretation Introduction
Interpretation:
The compound should be selected from the following which might be an ingredient in a commercial sunscreen:
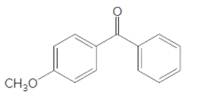
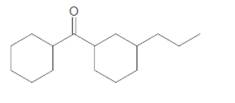
Concept Introduction:
Sunscreen ingredients are organic molecules which contains benzene rings within the molecule.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Mention Propranolol two most important medicinal uses.
What is an oxide of nitrogen that serves as an anesthetic and analgesic.
GIVE THE HOUSEHOLD NAME OF THIS
COMPOUND
Chapter 13 Solutions
General, Organic, and Biological Chemistry - 4th edition
Ch. 13.1 - Convert each condensed structure to a complete...Ch. 13.1 - Prob. 13.1PCh. 13.1 - Complete the structure of zingiberene, a component...Ch. 13.2 - Prob. 13.2PPCh. 13.2 - Prob. 13.3PCh. 13.2 - Prob. 13.3PPCh. 13.2 - Prob. 13.4PPCh. 13.3 - Prob. 13.5PPCh. 13.3 - Prob. 13.4PCh. 13.3 - Prob. 13.6PP
Ch. 13.3 - Prob. 13.5PCh. 13.3 - Prob. 13.6PCh. 13.3 - Prob. 13.7PCh. 13.3 - Prob. 13.8PCh. 13.6 - Prob. 13.7PPCh. 13.6 - Prob. 13.9PCh. 13.6 - What products is formed in each of the following...Ch. 13.6 - Prob. 13.9PPCh. 13.6 - What product is formed when l-pentene (CH3CH2CH2CH...Ch. 13.7 - Prob. 13.11PCh. 13.8 - Prob. 13.10PPCh. 13.8 - Prob. 13.12PCh. 13.8 - Prob. 13.13PCh. 13.10 - Prob. 13.11PPCh. 13.10 - Prob. 13.14PCh. 13.11 - Prob. 13.15PCh. 13.12 - Prob. 13.16PCh. 13.13 - Prob. 13.17PCh. 13.13 - Prob. 13.18PCh. 13 - Anethole, the major constituent of anise oil, is...Ch. 13 - Prob. 20PCh. 13 - What is the molecular formula for a hydrocarbon...Ch. 13 - Prob. 22PCh. 13 - Prob. 23PCh. 13 - Prob. 24PCh. 13 - Prob. 25PCh. 13 - Prob. 26PCh. 13 - Give the IUPAC name for each molecule depicted in...Ch. 13 - Give the IUPAC name for each molecule depicted in...Ch. 13 - Give the IUPAC name for each compound. a....Ch. 13 - Give the IUPAC name for each compound. d....Ch. 13 - Give the IUPAC name for each alkene.Ch. 13 - Give the I UP AC name for each alkene.Ch. 13 - Give the IUPAC name for each cyclic compound.Ch. 13 - Give the IUPAC name for each cyclic compound.Ch. 13 - Give the structure corresponding to each IUPAC...Ch. 13 - Prob. 36PCh. 13 - Each of the following IUPAC names is incorrect....Ch. 13 - Each of the following IUPAC names is incorrect....Ch. 13 - Prob. 39PCh. 13 - Prob. 40PCh. 13 - Prob. 41PCh. 13 - Prob. 42PCh. 13 - Prob. 43PCh. 13 - Label the carbon-carbon double bond as cis or...Ch. 13 - Prob. 45PCh. 13 - Prob. 46PCh. 13 - Prob. 47PCh. 13 - Prob. 48PCh. 13 - Prob. 49PCh. 13 - Prob. 50PCh. 13 - Prob. 51PCh. 13 - Prob. 52PCh. 13 - Prob. 53PCh. 13 - Prob. 54PCh. 13 - What alkyd halide is formed when each alkene is...Ch. 13 - Prob. 56PCh. 13 - Prob. 57PCh. 13 - Prob. 58PCh. 13 - What alkene is needed as a starting material to...Ch. 13 - Prob. 60PCh. 13 - Prob. 61PCh. 13 - Prob. 62PCh. 13 - Prob. 63PCh. 13 - Prob. 64PCh. 13 - Prob. 65PCh. 13 - Prob. 66PCh. 13 - Prob. 67PCh. 13 - Prob. 68PCh. 13 - Prob. 69PCh. 13 - Prob. 70PCh. 13 - Prob. 71PCh. 13 - Prob. 72PCh. 13 - Prob. 73PCh. 13 - Are o-bromochlorobenzene and m-bromochlorobenzene...Ch. 13 - Give the structure corresponding to each IUPAC...Ch. 13 - Give the structure corresponding to each IUPAC...Ch. 13 - Prob. 77PCh. 13 - Prob. 78PCh. 13 - Prob. 79PCh. 13 - Prob. 80PCh. 13 - Prob. 81PCh. 13 - Prob. 82PCh. 13 - Prob. 83PCh. 13 - Prob. 84PCh. 13 - Prob. 85PCh. 13 - Eleostearic acid is an unsaturated fatty acid...Ch. 13 - Prob. 87PCh. 13 - Prob. 88PCh. 13 - Prob. 89PCh. 13 - Prob. 90PCh. 13 - Prob. 91PCh. 13 - Prob. 92PCh. 13 - Prob. 93PCh. 13 - Prob. 94PCh. 13 - Answer the following questions about compound A,...Ch. 13 - Prob. 96PCh. 13 - Answer the following questions about alkene C,...Ch. 13 - Prob. 98PCh. 13 - Prob. 99PCh. 13 - Prob. 100PCh. 13 - Prob. 101CPCh. 13 - Prob. 102CP
Knowledge Booster
Similar questions
- Can napthalene dissolve in cooking oil?arrow_forwardAlkaloids are basic nitrogen-containing compounds of plant origin, many of which are physiologically active when administered to humans. Ingestion of coniine, isolated from water hemlock, can cause weakness, labored respiration, paralysis, and eventually death. Coniine is the toxic substance in the “poison hemlock” used in the death of Socrates. In small doses, nicotine is an addictive stimulant. In larger doses, it causes depression, nausea, and vomiting. In still larger doses, it is a deadly poison. Solutions of nicotine in water are used as insecticides. Cocaine is a central nervous system stimulant obtained from the leaves of the coca plant.Classify each amino group in these alkaloids according to type (primary, secondary, tertiary, aliphatic, aromatic, heterocyclic).arrow_forwardHow does the branching of hydrocarbon chain of synthetic detergents affect their biodegradability?arrow_forward
- Answer the following questions: 1. Why do liquids assume different shapes on the surface of the waxed paper? 2. What happens to the blade as it was placed on water, kerosene, and ethanol? Differentiate them. Why is this phenomenon possible? 3. What happens when the blade was placed on the detergent solution? What is the effect of the detergent on the IMFA of water?arrow_forwardEster compounds often have a sweet, pleasant odor. Many characteristic fruit scents are largely due to the natural presence of one or more ester compounds. As such, artificial scents for foods are often composed of complex mixtures of various esters. The exact identity and ratio of ingredients that compose a particular scent are closely guarded secrets in the food and fragrance industry. Suppose that you are a chemist working for a company that i creating a new line of air fresheners. The company is considering three scents: apple, pear, and pineapple. The project manager has asked you to prepare the ester compounds that are largely responsible for these scents. The structural formulas for these ester compounds are shown here: Alcohols for Air Freshener Project Molar mass Density Cost, per (g/mL) Reagent (g/mol) 1.00 L methanol 32.04 0.79 $46.20 ethanol 46.07 0.79 $112.00 1-propanol 60.10 0.80 $72.70 1-butanol 74.12 0.81 $72.60 Use the structural formulas of the alcohols and…arrow_forwardPropane can be used as a starting material for either propanal or propanone. Explain how this is possible. Provide a chemical equation for the reactions.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,


Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

General, Organic, and Biological Chemistry
Chemistry
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:Cengage Learning

Organic And Biological Chemistry
Chemistry
ISBN:9781305081079
Author:STOKER, H. Stephen (howard Stephen)
Publisher:Cengage Learning,