
Concept explainers
(a)
Interpretation:
The carbon atoms in the following structure should be classified.
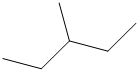
Concept Introduction:
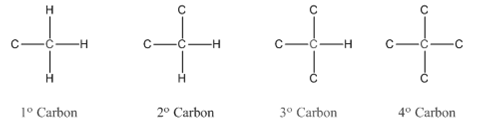
Compounds consist of carbon and hydrogen is known as hydrocarbons. Hydrocarbons are classified as saturated hydrocarbon and unsaturated hydrocarbon. Saturated hydrocarbons are those hydrocarbons in which carbon-carbon single bond is present as carbon is linked with four atoms.
In
Primary carbon (
Secondary carbon (
Tertiary carbon (
Quaternary carbon (
(b)
Interpretation:
The carbon atoms in the following structure should be classified.
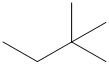
Concept Introduction:
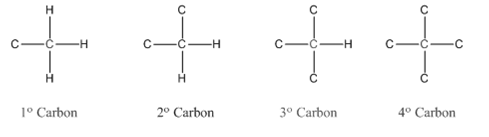
Compounds consist of carbon and hydrogen is known as hydrocarbons. Hydrocarbons are classified as saturated hydrocarbon and unsaturated hydrocarbon. Saturated hydrocarbons are those hydrocarbons in which carbon-carbon single bond is present as carbon is linked with four atoms. Unsaturated hydrocarbons are those hydrocarbons in which carbon-carbon multiple bonds are present that is double and triple bond.
In organic chemistry, carbons are classified as primary, secondary, tertiary and quaternary carbons.
Primary carbon (
Secondary carbon (
Tertiary carbon (
Quaternary carbon (
Want to see the full answer?
Check out a sample textbook solution
Chapter 12 Solutions
General, Organic, and Biological Chemistry - 4th edition
- Write the condensed structure for each of these skeletal structures. Cl condensed structure: condensed structure:arrow_forwardHow many electron pairs are shared when a triple bond exists between two carbon atoms? What must he the geometric arrangement around the carbon atoms in a triple bond? Draw the Lewis structure of a simple molecule that contains a triple bond.arrow_forwardSummarize the nomenclature rules for alkanes, alkenes, alkynes, and aromatic compounds. Correct the following false statements regarding nomenclature of hydrocarbons. a. The root name for a hydrocarbon is based on the shortest continuous chain of carbon atoms. b. The suffix used to name all hydrocarbons is -ane. c. Substituent groups are numbered so as to give the largest numbers possible. d. No number is required to indicate the positions of double or triple bonds in alkenes and alkynes. e. Substituent groups get the lowest number possible in alkenes and alkynes. f. The ortho- term in aromatic hydrocarbons indicates the presence of two substituent groups bonded to carbon- 1 and carbon-3 in benzene.arrow_forward
- I cannot understand where to put any of the figures for this skeletal structurearrow_forwardWrite condensed structural formulas, and skeletal structures for each structural isomer of C4H10.arrow_forwardI have given you a condensed structure. You need to convert it to an accurate bond-line structure. CH3 CH3-CH-CH2-CH-CH₂-C-H 12-CH-CH2₂-C-1 CI Draw (as bond-line structures) isomers of this compound where you only move the chlorine atom. Draw four isomers of the original compound that would have a five carbon chain as the longest chain. [Note: there would be many isomers that will satisfy this. Find any four.] Using the original molecule (and looking at the carbon next to the aldehyde carbonyl) what would be the charge on that carbon if I removed an H atom and left behind the pair of electrons? Circle the best answer Positive Negative Neutral In the space below, draw that structure (from the sentence above) as a bond-line structure. Then, draw a resonance structure for this ion and be sure to add curved arrows to show the movement of electrons.arrow_forward
 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning




