
Concept explainers
(a)
Interpretation:
The
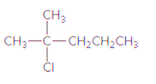
Concept Introduction:
The reactions in which alkane is reacted with halogen, like Cl2 or Br2 is known as halogenation reaction which results in the formation of alkyl halide and hydrogen halide. The important condition for this reaction to occur is heat or exposure to light.
(b)
Interpretation:
The alkane needed as a starting material should be determined which forms following alkyl halides:
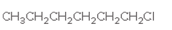
Concept Introduction:
The reactions in which alkane is reacted with halogen, like Cl2 or Br2 is known as halogenation reaction which results in the formation of alkyl halide and hydrogen halide. The important condition for this reaction to occur is heat or exposure to light.
Want to see the full answer?
Check out a sample textbook solution
Chapter 12 Solutions
General, Organic, and Biological Chemistry - 4th edition
- 22. Which of the following reactants will add symmetrically to alkenes? a. H20, H2SO4 O b. H2, Pt Ос. НС Od. HBrarrow_forwardwhat is the most stable alkene?arrow_forwardWhat functional group distinguishes each of the following hydrocarbon derivatives? a. halohydrocarbons b. alcohols c. ethers d. aldehydes e. ketones f. carboxylic acids g. esters h. amines Give examples of each functional group. What prefix or suffix is used to name each functional group? What are the bond angles in each? Describe the bonding in each functional group. What is the difference between a primary, secondary, and tertiary alcohol? For the functional groups in ah, when is a number required to indicate the position of the functional group? Carboxylic acids are often written as RCOOH. What does COOH indicate and what does R indicate? Aldehydes are sometimes written as RCHO. What does CHO indicate?arrow_forward
- Isooctane is the common name of the isomer of C8H18 used as the standard of 100 for the gasoline octane rating: (a) What is the IUPAC name for the compound? (b) Name the other isomers that contain a five-carbon chain with three methyl substituents.arrow_forwardIdentify the Type of Product 1-propene + H2O—> 1-propanol a. zaitsev b. markovnikov c. anti-markovnikov d. hoffmanarrow_forwardWhich of the following could be the molecular formula for an alkene? A. C7H7 B. C8H16 C. C2H3 D. C2H2 #. C2H6arrow_forward
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning





