
Concept explainers
(a)
Interpretation:
The number of C atoms present in the longest chain of the molecule shown below needs to be deduced.
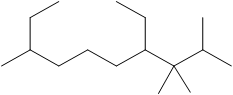
Concept Introduction:
(b)
Interpretation:
The number of C atoms present in the longest chain of the molecule shown below needs to be deduced.
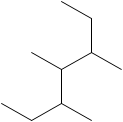
Concept Introduction:
Alkanes have a general formula, CnH2n+2where n denotes the number of C atoms. In the case of linear or branched alkanes the parent chain is the one which has the highest number of carbon atoms. The longest continuous chain of C atoms forms the base name of the compound.
Want to see the full answer?
Check out a sample textbook solution
Chapter 12 Solutions
General, Organic, and Biological Chemistry - 4th edition
- How many electron pairs are shared when a triple bond exists between two carbon atoms? What must he the geometric arrangement around the carbon atoms in a triple bond? Draw the Lewis structure of a simple molecule that contains a triple bond.arrow_forward= ORGANIC CHEMISTRY Drawing a skeletal structure from a condensed structure Draw a skeletal ("line") structure of this molecule: OH CH3 -C-CH₂-CH=CH₂ OHarrow_forwardDraw the condensed structure of an isomer of this molecule: OH CH₂ C-CH₂-OHarrow_forward
- What is the difference between a structural isomer and structural conformations of an organic molecule?arrow_forwardDraw the Lewis structure of CH3CH₂CH₂CH₂CH₂CN. Include all lone pairs and charges as appropriate.arrow_forwardDraw the structure of compound C4H9COCH3C4H9COCH3 to show all individual atoms and all covalent bonds. Assume that all carbons are connected in a continuous chain.arrow_forward
- With reference to organic molecules, explain the following structure: CH2 CH— О CH2arrow_forward12. Identify (adraw a circle and label the class) the classes of organic molecules represented in the following molecule Ноarrow_forwardExplain general structure of Compounds Containing a C=O Group ?arrow_forward
 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning

