
Study Guide for Campbell Biology
11th Edition
ISBN: 9780134443775
Author: Lisa A. Urry, Michael L. Cain, Steven A. Wasserman, Peter V. Minorsky, Jane B. Reece, Martha R. Taylor, Michael A. Pollock
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 9, Problem 2IQ
Fill in the appropriate terms in the following equation.
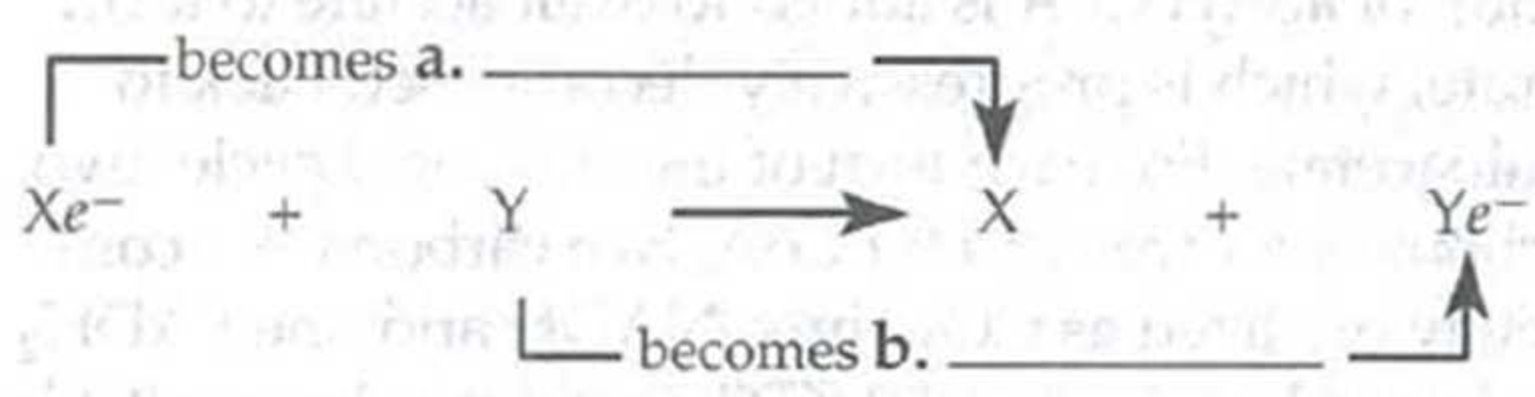
Xe– is the reducing agent; it c. _____ electrons.
Y is the d. _____; it e. _____ electrons.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Identify the following electron transport intermediate. H : O : O : HA. hydroxyl free radical B. the superoxide free radicalC. hydroxide ionD. hydrogen peroxideE. water
-n
f
estion
B
I
ut of
question
PF
budy
As you increase the amount of substrate in a reaction (while keeping the enzyme concentration the same):
Select one:
O A. The amount of products formed should decrease
OB. The amount of products formed should remain the same
OC. The amount of products formed should increase
Clear my choice
Mixing hydrogen peroxide with an enzyme different than catalase (such as lactase) should also result in the formation of products.
Select one:
O True
O False
The temperature at which an enzyme works best can differ from enzyme to enzyme.
Select one:
O True
False
B
7
O
Which of the followingdescribe superior properties of enzymes (biological catalysts) over traditional chemical catalysts?
a. They are mostly and generally operative under mild temperature, pressure, and pH conditions
b. They are regulated only by substrate concentration
c. They do not effect the reaction equilibrium, but lower the reaction's activation energy
d. They are recycled at the end of the reaction
Choose all that apply
Chapter 9 Solutions
Study Guide for Campbell Biology
Ch. 9 - Fill in the following summary equation for...Ch. 9 - Fill in the appropriate terms in the following...Ch. 9 - a. In the conversion of glucose and O2 to CO2 and...Ch. 9 - a. NAD+ is called a(n) ____________________. b....Ch. 9 - Fill in the three stages of cellular respiration...Ch. 9 - Fill in the blanks in the following summary...Ch. 9 - Fill in the blanks in the following diagram of the...Ch. 9 - Label the following diagram of oxidative...Ch. 9 - Fill in the following tally for the maximum ATP...Ch. 9 - How much more ATP can be generated by aerobic...
Ch. 9 - This chapter describes how catabolic pathways...Ch. 9 - Prob. 2SYKCh. 9 - Fill in the following table to summarize...Ch. 9 - When electrons move closer to a more...Ch. 9 - In the reaction C6H12O6 + 6 O2 6 CO2 + 6 H2O, a....Ch. 9 - In which of the following conversions is the first...Ch. 9 - Some prokaryotes use anaerobic respiration, a...Ch. 9 - Which of the following reactions is incorrectly...Ch. 9 - In which organelle of a plant cell does the citric...Ch. 9 - Which of the following compounds produces the most...Ch. 9 - Aerobic eukaryotes produce CO2 as a by-product...Ch. 9 - Which of the following statements correctly...Ch. 9 - In the electron transport system, H+ ions are...Ch. 9 - When glucose is oxidized to CO2 and water,...Ch. 9 - The energy required for the chemiosmotic synthesis...Ch. 9 - Which of the following statements correctly...Ch. 9 - The citric acid cycle is best described as a. the...Ch. 9 - Fermentation produces less ATP than cellular...Ch. 9 - If muscle cells do not receive enough oxygen from...Ch. 9 - Glucose made from six radioactively labeled carbon...Ch. 9 - Glycolysis is considered one of the first...Ch. 9 - Which of the following substances produces the...Ch. 9 - Fats and proteins can be used as fuel in the cell...Ch. 9 - Which of the following statements is false...Ch. 9 - Brown fat, which is found in newborn infants and...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Similar questions
- Which of the following is incorrect about enzyme cofactors? a. Some can be metal ions b. None; all the other choices are correct O c. The so-called co-substrates are needed in equal [substrates] and they are not recycled Od. Some are called coenzymes which are classified into co-substrates and prosthetic groups +arrow_forwardAnaerobic digester reactors (technology) are used in small South African wastewater treatment works. The following reactors are very likely to take place in the type of reactorC-compounds + bacteria=XX + bacteria =CO2 + NH3a. Predict the products (X) if the above reactions occur? justify your answerb. What types of bacteria are likely to be present in the reactorsc. What could C compound represent for bacteria2.2 Predict the implication of operating above 60°C in the digester and reactorsarrow_forwardQuestion 23 An enzyme has a single active site at which it can bind and hydrolyze either X or Y but the enzyme cannot bind X and Y at the same time. Which of the following statements are TRUE? Multiple answers: Multiple answers are accepted for this question Select one or more answers and submit. For keyboard navigation... SHOW MORE V The Km for X will be affected if Y is present in the reaction mixture. a Y is a competitive inhibitor of X. The Km for X will increase. d. The V for X will be affected if Y is present in the reaction mixture. max pH dependence of Vmax reflects the ionization state of catalytic site e residues. Consider the following: X and Y are methanol (poisonous) and ethanol respectively. If the Km for X = 0.01 M and the Km for Y = 0.001 M then 0.01 M Y is 10 times the concentration of Y required for 0.5 Vmax. Addition of an enzyme to a chemical reaction increases the ratio of g products to reactants (Keg).arrow_forward
- Match the following catalytic strategies with their example. Place a Letter on the picture. There are only two examples given. CH2OH CH2OH CH2OH CH2OH он он OH OH + H20 OH OH OH но H. но но OH OH OH OH OH OH A. Oxidoreducatase B. Transferase C. Hydrolase D. Lyase NH3 E. Isomerase F. Ligase R-CH-COO "ooc-CH2-CH2-C-COO Amino acid a-Ketoglutaratearrow_forwardIn the process of isolating the enzyme amylase, you used methanol: To dissolve the starch O a. O b. To precipitate the non-polar enzyme O c. To dissolve the sugar O d. To help the enzyme catalyze the reaction. Oe. To precipitate the polar enzyme CLEAR MY CHOICEarrow_forwardIn order for the reaction rate to rise the activation energy should be Select one: O O O O a. decreased b. increased c. decreased or increased d. not changed Riboflavin gives rise to cofactors:arrow_forward
- Use the graph below to determine which of the following is the optimal temperature for catalase. Catalase Activity 1.5 1.4 1.3 1.2 1.1 1.0 0.9 0.8 0.7 0.6 0.5 0.4 0.3 0.2 0.1 10 20 30 40 50 60 Temperature (°C) O 0.8 O 1.45 O 37 O 20 Percent Increase in Oxygen per Minutearrow_forwardMatch the following catalytic strategies with their example. Place a Letter on the picture. There are only two examples given. CH2OH CH2OH H. CH,OH CH2OH он он OH Kon + H2O OH HO OH OH OH HO но OH OH OH OH A. Oxidoreducatase OH OH B. Transferase C. Hydrolase D. Lyase NH3 E. Isomerase F. Ligase R-CH-COO + 00C-CH2-CH2-C-COO Amino acid a-Ketoglutarate NH3 + 00c-CH2-CH2–CH-C0 R-C-COO a-Keto acid Glutamatearrow_forwardA. Build a graph (you can use Excel T) with the information shown in the table below (concentration x axis): CARBON SOURCE Transport rate (umol/min) CONCENTRATION COMPOUND COMPOUND (mM) 0.1 2.0 18 0.3 6.0 46 1.0 20 100 3.0 30 150 10.0 200 182 B. Determine from your graph whether the data for compound A corresponds to ethanol or acetate. |arrow_forward
- HOT Metabolite Substrate Product ATP ADP Pi NAD+ NADH FAD FADH₂ OH Concentration (mm) 0.318 0.224 4.95 0.825 0.724 2.98 1.73 1.42 0.843 HO HO Use the metabolite concentrations in the table to calculate the AG of the reaction above. Report your answer in kJ/mol to 3 significant figures. OHarrow_forwardIn a hydrogen fuel cell, hydrogen gas and oxygen gas are combined to form water. Write the balanced chemical equation describing this reaction using the lowest whole-number coefficients. chemical equation: oxygen hydrogen Identify the oxidizing agent. water H₂ + O₂ → H₂O water Incorrect Identify the reducing agent. hydrogen oxygen Determine the number of electrons transferred in the balanced chemical equation. number of electrons transferred: Incorrectarrow_forwardEnzymes act as catalysts in biochemical reactions. Reaction Model D.O. WX WX W In the diagram, which represents a catalyst? O W OY O WXarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON
Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education,
Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education, Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company
Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co.
Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co. Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education
Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education

Human Anatomy & Physiology (11th Edition)
Biology
ISBN:9780134580999
Author:Elaine N. Marieb, Katja N. Hoehn
Publisher:PEARSON

Biology 2e
Biology
ISBN:9781947172517
Author:Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:OpenStax

Anatomy & Physiology
Biology
ISBN:9781259398629
Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa Stouter
Publisher:Mcgraw Hill Education,

Molecular Biology of the Cell (Sixth Edition)
Biology
ISBN:9780815344322
Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter Walter
Publisher:W. W. Norton & Company

Laboratory Manual For Human Anatomy & Physiology
Biology
ISBN:9781260159363
Author:Martin, Terry R., Prentice-craver, Cynthia
Publisher:McGraw-Hill Publishing Co.

Inquiry Into Life (16th Edition)
Biology
ISBN:9781260231700
Author:Sylvia S. Mader, Michael Windelspecht
Publisher:McGraw Hill Education
Biochemical Tests-Part 1; Author: Southern Stacker;https://www.youtube.com/watch?v=a-i9vANfQWQ;License: Standard Youtube License