
Interpretation:
The conditions required for ionic compounds to conduct electricity and reason for it not being used as conductor always needs to be explained.
Concept introduction:
Molecules are bound together by different types of bonds namely ionic and covalent. Ionic bonds are formed when there is complete transfer of electrons from one atom to another. These atoms either lose or gain electrons to become negatively or positively charged ions. The forces of attraction between these ions causes the ionic bond formation. When molten, the ionic compounds will dissociate to release ions which are free to move around the semi liquid state and hence will be able to conduct electricity.
Answer to Problem 72A
The ionic compound will conduct electricity only in molten form or when dissolved in water.
Explanation of Solution
Ionic bonds involve the transfer of electrons from one atom to the other. The metal atoms loses electrons to form a positively charged ion while the non-metal atoms gains electrons to form negatively charged ions. During the process of donating or gaining electrons, the atoms follow octet rule to attain a stable noble gas configuration.
The ionic compound is a solid but when heated the bonds holding the ions are free to move within the structure. Also, when the solid is dissolved in water, it will break free the lattice points. The ions held at the lattice points are dissociated to free ions. Now, when current is passed through the ions, it will carry the charges. Hence, when electricity is passed through molten or solution containing ionic solids, it is found to conduct electricity.
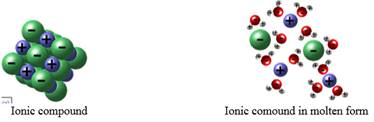
Therefore, ionic compounds will conduct electricity only in molten form or when dissolved in water.
The ionic compound will conduct electricity only in molten form or when dissolved in water.
Chapter 7 Solutions
Chemistry: Matter and Change
Additional Science Textbook Solutions
Introductory Chemistry (5th Edition) (Standalone Book)
Chemistry: Structure and Properties (2nd Edition)
Organic Chemistry (9th Edition)
General Chemistry: Principles and Modern Applications (11th Edition)
CHEMISTRY-TEXT
Chemistry: The Central Science (13th Edition)
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





