
Principles of Modern Chemistry
8th Edition
ISBN: 9781305079113
Author: David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
Chapter 7, Problem 48CP
Interpretation Introduction
Interpretation: Whether the wavelength of maximum absorption in naphthalene is shorter or longer than 255 nm that is maximum absorption in benzene needs to be determined.
Concept Introduction:Benzene shows resonance due to delocalization of
electrons. The naphthalene is formed by fusion of two benzene molecules. The structure of benzene and naphthalene is represented as follows:
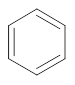 and
and 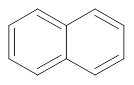
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
what is the term used to describe organic reactions in which each atom of a diatomic molecule is transferred to one of the carbons in a double bond?
In an advanced analytical chemistry lab, a team analyzing a compound 'Q' known to be a structural isomer of octane (C8H18). To determine the specific structure of 'Q', a series of spectroscopic
analyses are performed. The sequence of the analysis involves: Infrared (IR) spectroscopy, which indicates the absence of functional groups like alcohols, ketones, and carboxylic acids. Nuclear
Magnetic Resonance (NMR) spectroscopy, showing signals indicative of only methyl and methylene groups, with no evidence of methine (CH) or quaternary carbon environments. Mass spectrometry
(MS), revealing a fragmentation pattern consistent with branched alkane structures. Based on this sequence of analyses, what is the most likely structure of compound 'Q'? Options: A. 2,2,4-
Trimethylpentane B. n-Octane C. 2-Methylheptane D. 3-Ethylhexane Don't use chatgpt please provide valuable answer
What are the hybridization and bond angles of the oxygens in benzoic acid?
Chapter 7 Solutions
Principles of Modern Chemistry
Ch. 7 - Prob. 1PCh. 7 - Is it possible for a motor fuel to have a negative...Ch. 7 - A gaseous alkane is burned completely in oxygen....Ch. 7 - A gaseous alkyne is burned completely in oxygen....Ch. 7 - Write a chemical equation involving structural...Ch. 7 - Prob. 6PCh. 7 - Prob. 7PCh. 7 - Prob. 8PCh. 7 - Prob. 9PCh. 7 - Prob. 10P
Ch. 7 - Prob. 11PCh. 7 - Prob. 12PCh. 7 - Prob. 13PCh. 7 - State the hybridization of each of the carbon...Ch. 7 - Prob. 15PCh. 7 - Prob. 16PCh. 7 - Prob. 17PCh. 7 - In a recent year, the United States produced...Ch. 7 - Prob. 19PCh. 7 - Prob. 20PCh. 7 - Prob. 21PCh. 7 - Prob. 22PCh. 7 - Prob. 23PCh. 7 - Prob. 24PCh. 7 - Prob. 25PCh. 7 - Prob. 26PCh. 7 - Acetic acid can be made by the oxidation of...Ch. 7 - Acrylic fibers are polymers made from a starting...Ch. 7 - Compare the bonding in formic acid (HCOOH) with...Ch. 7 - Prob. 30PCh. 7 - Prob. 31PCh. 7 - Prob. 32PCh. 7 - Prob. 33PCh. 7 - Prob. 34PCh. 7 - Prob. 35PCh. 7 - Describe the changes in hydrocarbon structure and...Ch. 7 - trans-Cyclodecene boils at 193C, but...Ch. 7 - Prob. 38APCh. 7 - Prob. 39APCh. 7 - Consider the following proposed structures for...Ch. 7 - Prob. 41APCh. 7 - Prob. 42APCh. 7 - Prob. 43APCh. 7 - Prob. 44APCh. 7 - Prob. 45APCh. 7 - The steroid stanolone is an androgenic steroid (a...Ch. 7 - The structure of the molecule cyclohexene is Does...Ch. 7 - Prob. 48CP
Knowledge Booster
Similar questions
- The structure of the molecule cyclohexene is Does the absorption of ultraviolet light by cyclohexene occur at longer or at shorter wavelengths than the absorption by benzene? Explain.arrow_forwardIn a time-resolved picosecond spectroscopy experiment, Sheps, Crowther, Carrier, and Crim (Journal of Physical Chemistry A, Vol. 110, 2006; pp. 30873092) generated chlorine atoms in the presence of pentane. The pentane was dissolved in dichloromethane, CH2C12. The chlorine atoms are free radicals and are very reactive. After a nanosecond the chlorine atoms have reacted with pentane molecules, removing a hydrogen atom to form HCl and leaving behind a pentane radical with a single unpaired electron. The equation is Cl (dcm) + C5H12(dcm) HCl(dcm) + C5H11 (dcm) where (dcm) indicates that a substance is dissolved in dichloromethane. Measurements of the concentration of chlorine atoms were made as a function of time at three different concentrations of pentane in the dichloromethane. These results are shown in the table. (a) Determine the order of the reaction with respect to chlorine. (b) Determine whether the reaction rate depends on the concentration of pentane in dichloromethane. If so, determine the order of the reaction with respect to pentane. (c) Explain why the concentration of pentane in dichloromethane does not affect the data analysis that you performed in part (a). (d) Write the rate law for the reaction and calculate the rate of reaction for a concentration of chlorine atoms equal to 1.0 M and a pentane concentration of 0.23 M. (e) Sheps, Crowther, Carrier, and Crim found that the rate of formation of HCl matched the rate of disappearance of Cl. From this they concluded that there were no intermediates and side reactions were not important. Explain the basis for this conclusion.arrow_forwardDraw Lewis structures and condensed structural formulas for the four alcohols with the molecular formula C4H10O. Classify each alcohol as primary, secondary, or tertiary. (Hint: First consider the connectivity of the four carbon atoms; they can be bonded either four in a chain or three in a chain with the fourth carbon as a branch on the middle carbon. Then consider the points at which the iOH group can be bonded to each carbon chain.)arrow_forward
- You were tasked to propose a structure for a compound with a chemical formula of C₂H₂N. In the laboratory, the compound appears as a clear, colorless liquid. To determine the structure of the compound, you subject the compound to several solubility tests and chemical tests. The observations for these tests are summarized: SOLUBILITY GROUP: Group V Insoluble in water and 10% NaOH but soluble in 10% HCI FUNCTIONAL GROUP/CLASS: Amine 1) Based on the results of the solubility tests and chemical tests, what is the most probable structure of C7H9N? Draw your answer in the box below. You may draw the compound using line-bond formula OR Lewis structure. |arrow_forwardWhich of the following is FALSE about the absorbance of a compound? O A. Organic solvents have effect on the absorbance of the compounds. B. Organic compounds with increasing conjugation will absorb UV-Vis radiation C. Saturated hydrocarbons will not give an absorbance reading. D. Solutions containing transition metals are not capable to absorb UV-Vis radiation.arrow_forwardGlucose, C6H12O6, contains an aldehyde group but exist predominantly in the form of the cyclic hemiacetal show below. A cyclic hemiacetal is formed when the —OH group of one carbon bonds to the carbonyl group of another carbon. Identify which carbon provides the —OH group and which provides the —CHO? Give a functional isomer of glucose and draw its structure.arrow_forward
- Why does the hybridization of the nitrogen atom in the pyrrole and pyrrolidine molecules have to be different? Why might it look the same at first glance? Please explain.arrow_forwardConsider the compound 2,4-dimethyl-3-isopropylpentane. How many unique radical structures can be derived from this hydrocarbon when it undergoes free radical substitution? Write the number of free radicals formed.arrow_forwardExplain the Infrared Spectra of Common Functional Groups ?arrow_forward
- Identify and label all functional groups (alcohols, amines, amides, carboxylic acids, ketones, aldehydes, aromatic rings, aromatic amines, etc) of Carvone on the Lewis Structure. You do not have to circle alkanes. Make sure to add additional details about the functional groups of Carvonearrow_forwardThere are three ethers with he formula C4H10O. Draw their structures.arrow_forwardThe five parts of question 16 relate to the following three molecules: N-H А в с A (a) Which of the three molecules above are structural isomers? (b) Which of the molecules contains a carbon with linear geometry? (c) Which molecule contains a tertiary amine? (d) Which molecule only contains sp³ hybridized atoms (not including hydrogen)? (e) Which molecule is chiral? Circle the stereocentre in this molecule.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning