
Concept explainers
(a)
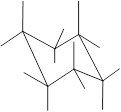
Interpretation: The name of the given structure needs to be determined.
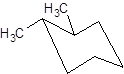
Concept Introduction: Chair conformation is the most stable conformation of cyclohexane. It is represented as follows:

Here, the substituted groups in the chair conformation are represented as follows:
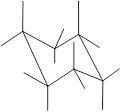
The groups showing in straight upward and downward directions are axial and the groups bend slightly right or left are equatorial.
The axial and equatorial groups are represented in the chair conformation as A and E:
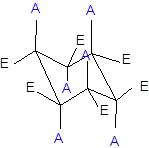
(b)
Interpretation: The given structure on the right needs to be completed which is obtained by chair flip.
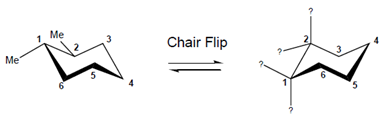
Concept Introduction:After a chair flip, all the axial bonds become equatorial and all the equatorial bonds become axial. But, the groups in the up direction remains upward and groups in down direction remain downward.
Also, if the molecule is cis, it remains cis even after chair flip. The same is the case with trans molecule.
(c)
Interpretation: The name of the structure formed after the chair flip in part (b) needs to be determined.
Concept Introduction: Chair conformation is the most stable conformation of cyclohexane. It is represented as follows:

Here, the substituted groups in the chair conformation are represented as follows:
The groups showing in straight upward and downward directions are axial and the groups bend slightly right or left are equatorial.
The axial and equatorial groups are represented in the chair conformation as A and E:
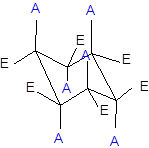
(d)
Interpretation: Whether the two structures are consistent with the given facts about chair flip or not needs to be determined.
Concept Introduction:
The chair conformation of cyclohexane is represented as follows:

During the flipping, no bond is break. The numbering in the chair form is represented as follows:
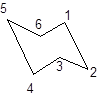
During ring flipping, mirror image of the chair conformation is formed.
It is represented as follows:
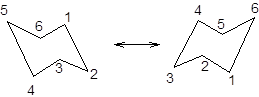
Trending nowThis is a popular solution!

Chapter 7 Solutions
Organic Chemistry: A Guided Inquiry
- A student suggests that the molecule on the right can be made from a single molecule with one less ring in it. If the student is correct, draw the starting material below, otherwise, check the box under the drawing area. ? + NaOH Click and drag to start drawing a structure. olo Ararrow_forward1. Draw ethylbenzene and put a + on the carbon next to the benzene (innermost carbon in ethyl group). Show all resonance forms for this ion. 2. draw the naphthalene and put one - on the neighboring carbon of the "crossroads". show every resonance forms for this ion. 3. Draw benzoic acid and draw on all p-orbitals.arrow_forwardModified True or False. Write correct if the statement is True and if false write the word/s that make it false and beside it write the word/s that will make the statement true. 4. Lone pair delocalization decreases the positivity of carbonyl carbon.5. The longer the carbon chain, the higher the boiling point.6. sp3 is 75% p character and this allows C-H hyper conjugation.7. The higher the electronegativity, the higher the temperature needed to break bonds.8. Steric effect increases the boiling point.9. Stearic effect increases the Van der Waals forces.10. Lone pair delocalization decreases the positivity of carbonyl carbon.11. Amines are considered basic.12. The stronger the Van der Waals forces between molecules of the same substance, themore soluble the substance is in water.13. The longer the carbon chain, the lower the solubility of the substance in water.14. Alkanes with less than 6 carbon atoms are gases.15. Stearic effects enhances the solubility in water.16. H-bond can exist…arrow_forward
- a • You should include all products. • Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right corner. Separate multiple products using the + sign from the drop-down menu. H ? + H₂O H* Sn [Farrow_forwardFor each resonance structure, use curved arrows to show the delocalization of electron pairs that transforms the structure into the indicated resonance structure. : NH2 NH2 NH2 NH, resonance resonance resonance resonance structure A structure B structure C structure D Do not delete any pre-drawn bonds, charges, or lone pairs. If you accidentally delete a vital part of the structure, use the undo button on the lower left of the drawing canvas. 1. Draw curved arrows to form structure B. 2. Draw curved arrows to form structure C. Select Draw Rings More Erase Select Draw Rings More Erase C C H Narrow_forward20. Circle any two structures. Below each structure, write the name of the compound. Onl graded if you draw more than two. Use cis or trans in the name.arrow_forward
- Circle the correct “Class” name for each pair.1. Aromatic or benzene ring2. Hydroxyl or alcohol3. Single bond or Alkane4. Thiol or sulfhydryl5. Carbonyl or Aldehyde Circle the correct “Functional group” name for each pair6. Triple bond or Alkyne7. Alcohol or hydroxyl8. Amine or Amino9. Carbonyl or Ketone10. Ether or Alkoxyarrow_forwardFirst, add curved arrow(s) to show the resonance using the following pattern: an allylic carbocation. Modify the second structure given to draw the new resonance structure. Include lone pairs and charges in your structure. Use the + and - tools to add charges to an atom, and use the single bond tool to add/remove double bonds. .CH3 .CH3 ČH3arrow_forwardFor each resonance structure, use curved arrows to show the delocalization of electron pairs that transforms the structure into the indicated resonance structure. + + : NH2 NH2 NH2 NH2 resonance resonance resonance resonance structure A structure B structure C structure D Do not delete any pre-drawn bonds, charges, or lone pairs. If you accidentally delete a vital part of the structure, use the undo button on the lower left of the drawing canvas.arrow_forward
- OH a. Draw the cis and trans isomer of the molecule above. b. Draw the chair form of the trans isomer of the molecule. C. Draw the flipped chair form of the trans isomer of the molecule. d. What is the more stable chair form?arrow_forwardQUESTION 2 Label the stereocenters (C and CA) as R or S, and then choose the correct name for Molecule W. (Note: R and S designations are placed in front of the name in parentheses. The number in front of the R or S indicates where the stereocenter appears in the base chain.) CI C2- Molecule W O A. (2R,4R)-2-chloro-4-fluorohexane O B. (2R,4S)-2-chloro-4-fluorohexane O C. (2S,4R)-2-chloro-4-fluorohexane O D. (2S,4S)-2-chloro-4-fluorohexanearrow_forwardDraw the simplest possible set of curved arrows that shows how the structure on the left could be turned into the structure on the right. H. Br. H H- H. H. H. Harrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
