
Organic Chemistry: A Guided Inquiry
2nd Edition
ISBN: 9780618974122
Author: Andrei Straumanis
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 6, Problem 34CTQ
Label each double bond E, Z, or neither. (It may help to draw in some missing H’s.)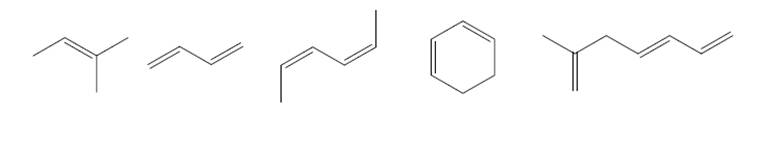
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Draw curved arrows to illustrate the bond-making and bond-breaking processes.
Draw all missing reactants and/or products in the appropriate boxes by placing atoms on the canvas and connecting them with bonds.
arrows should start on an atom or a bond and should end on an atom, bond, or location where a new bond should be created.
D
M
10
+
[1]
H₂C
י
H EXP. CONT.
L
1
CH₂
HBr:
d
H₂C
CH3
H
C
N
O
S
CI
Br
I
P
F
Curved arrows are used to illustrate the flow of electrons. Using the provided
resonance structures, draw the curved electron-pushing arrows to show the
interconversion between resonance hybrid contributors.
Be sure to account for all bond-breaking and bond-making steps.
Drawing Arrows
N
▸
Q
Rank in order of decreasing strength of the indicated bond.
(1 = strongest bond; 4 = weakest bond)
B
D
D
Chapter 6 Solutions
Organic Chemistry: A Guided Inquiry
Ch. 6 - Prob. 1CTQCh. 6 - Prob. 2CTQCh. 6 - Prob. 4CTQCh. 6 - Prob. 5CTQCh. 6 - Complete this graph of relative potential energy...Ch. 6 - Prob. 7CTQCh. 6 - Prob. 8CTQCh. 6 - Prob. 9CTQCh. 6 - Consider the Newman projection below. a. Draw a...Ch. 6 - Draw a Newman projection showing the lowest P.E....
Ch. 6 - Prob. 12CTQCh. 6 - Prob. 13CTQCh. 6 - In skeletal representations the hydrogens are not...Ch. 6 - Prob. 15CTQCh. 6 - Prob. 16CTQCh. 6 - Prob. 17CTQCh. 6 - Prob. 19CTQCh. 6 - Prob. 20CTQCh. 6 - Prob. 21CTQCh. 6 - Prob. 22CTQCh. 6 - Prob. 23CTQCh. 6 - Draw a constitutional isomer of pentane,...Ch. 6 - How many H’s are lost from the molecular formula...Ch. 6 - How many ifs are lost from the molecular formula...Ch. 6 - Prob. 27CTQCh. 6 - What is the degree of unsaturation for the example...Ch. 6 - Without counting hydrogens, determine which one of...Ch. 6 - Determine the degree of unsaturation (and draw a...Ch. 6 - a model of each molecule shown above: Is the...Ch. 6 - Prob. 32CTQCh. 6 - Prob. 33CTQCh. 6 - Label each double bond E, Z, or neither. (It may...Ch. 6 - Prob. 35CTQCh. 6 - Prob. 36CTQCh. 6 - Indicate the relationship between each pair....Ch. 6 - Prob. 38CTQCh. 6 - Prob. 1ECh. 6 - Prob. 2ECh. 6 - Using your model of butane (CH3CH2CH2CH3) ,...Ch. 6 - Consider the molecule 1-bromo-2-methylbutane. C3...Ch. 6 - Prob. 5ECh. 6 - Prob. 8ECh. 6 - Prob. 9ECh. 6 - Prob. 10ECh. 6 - Prob. 11ECh. 6 - Prob. 12ECh. 6 - Prob. 13ECh. 6 - Prob. 15ECh. 6 - Prob. 16ECh. 6 - Prob. 17ECh. 6 - Prob. 18ECh. 6 - Prob. 19ECh. 6 - Prob. 20ECh. 6 - Prob. 21ECh. 6 - Double bonds do not rotate freely under normal...Ch. 6 - up an example (not appearing in this ChemActivity)...Ch. 6 - Prob. 24ECh. 6 - Prob. 25E
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Curved arrows are used to illustrate the flow of electrons. Using the provided starting and product structures, draw the curved electron-pushing arrows for the following reaction or mechanistic steps. Be sure to account for all bond-breaking and bond-making steps. :OH: Select to Add Arrows H₂O 3 H₂ H HH H :Br:Ⓒ HH Select to Add Arrows H₂O 17 :Br: H H Please select a drawing or reagent from the question areaarrow_forwardx-xo B Draw molecule A. On that drawing include the lone pairs and the curved arrows that would produce resonance structure B.arrow_forward4. Draw the possible resonance structures for compounds C and D. Include lone pairs and indicate non-zero formal charges. Circle the minor resonance contributor for compound C. O :O: :O: C & Darrow_forward
- For the following, use curved arrows to show the transformation of one resonance structure into another. Label the major and minor resonance forms (major and minor contributors to the resonance hybrid), and show which ones are of equal energy. Briefly justify your choices. Draw the structure of the resonance hybrid. [re % la] uarrow_forwardP3A. Draw the resonance structures for the following compound and circle the one that contributes most to the resonance hybrid. Why? P3B. Draw the resonance structures for the following compound and circle the one that contributes most to the resonance hybrid. Why? dide animato lahompoto Inies anarrow_forwardFor 1 and 2, use curved arrows to illustrate the potential overall electron movements or bond changes, and identify the type of reaction by examining the overall chemical transformation. Show A-H bonds as needed.arrow_forward
- What orbitals are used to form each indicated bond in the molecule shown? a H C barrow_forward3. One resonance form of anion C,H¸O is drawn below. Draw one other resonance form of C,H,O', and indicate which of the two forms is more stable, explain your choice. H2C=ċ-CH3arrow_forward8. If a resonance structure must have a negative formal charge, that charge is most stable on an electronegative atom. Draw all relevant resonance structures of the molecule below with curved arrows only using pattern 1. Circle the most stable structure(s). Explain the choice of most stable structure with 1-3 complete sentences.arrow_forward
- t The chem3D image below is that of a cation; double bonds are missing. Draw one resonance stucture for the cation. The structure you draw should be a major contributor to the overall structure of the hybrid. ball & stick ♥ S + labels • You do not have to consider stereochemistry. • You do not have to explicitly draw H atoms. • You do not have to include lone pairs in your answer. • In cases where there is more than one answer, just draw one. [ ] در ? Previousarrow_forwardQ9. Circle the most basic nitrogen(s) on the molecule shown below. HN NH Piccarrow_forwardPhosphorus trichloride (PCl3) can be used to convert carboxylic acids into acid chlorides. The mechanism for the reaction is shown but the curved arrows and the formal charges on the respective atoms have been left out. a. Draw the missing curved arrows for each step. b. Add any missing formal chargesarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning
Mass Spectrometry; Author: Professor Dave Explains;https://www.youtube.com/watch?v=hSirWciIvSg;License: Standard YouTube License, CC-BY