
Organic Chemistry: A Guided Inquiry
2nd Edition
ISBN: 9780618974122
Author: Andrei Straumanis
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 6, Problem 17CTQ
Interpretation Introduction
Interpretation:
The common thing in the molecules given in Column I, II, and III in Model 7 should be predicted.
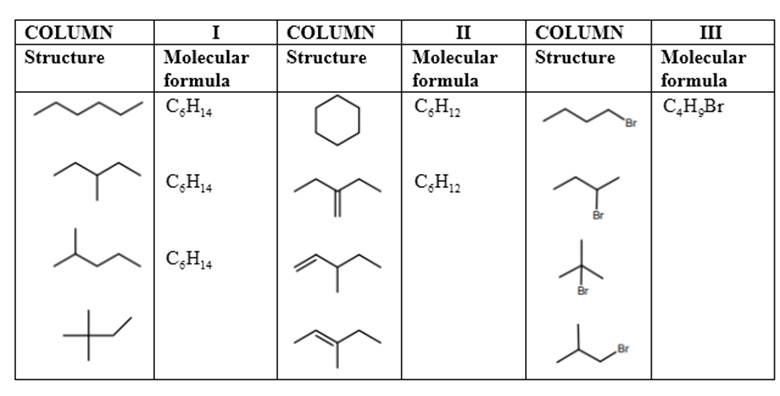
Concept Introduction:
Chemical formula or molecular formula represents the type of
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
10) Can H3PO4 exist with H₂PO4 in a mixture?
What about H₂PO4 and HPO4²?
What about H₂PO4 and PO,³?
What is the ratio of CATION to ANION in Drug X? [blank1]
H₂C
-H
Fogy
CH3 SO42
'N
CH3
Drug X
CH3NH
Describe the possible interactions between the SIDE CHAINS of amino acids:
I. and II....
II. and III...
I. and III...
1.
COO
NH3*
2
HO
II.
COO HO
NH3*
COO
NH3*
III.
Complete the following table. In the second column, assign each set of molecules as constitutional isomers, conformational isomers, enantiomers, diastereomers, or identical. For the third column, based on that analysis, indicate for each set of molecules if they are separable or inseparable by SiO2 chromatography.
Chapter 6 Solutions
Organic Chemistry: A Guided Inquiry
Ch. 6 - Prob. 1CTQCh. 6 - Prob. 2CTQCh. 6 - Prob. 4CTQCh. 6 - Prob. 5CTQCh. 6 - Complete this graph of relative potential energy...Ch. 6 - Prob. 7CTQCh. 6 - Prob. 8CTQCh. 6 - Prob. 9CTQCh. 6 - Consider the Newman projection below. a. Draw a...Ch. 6 - Draw a Newman projection showing the lowest P.E....
Ch. 6 - Prob. 12CTQCh. 6 - Prob. 13CTQCh. 6 - In skeletal representations the hydrogens are not...Ch. 6 - Prob. 15CTQCh. 6 - Prob. 16CTQCh. 6 - Prob. 17CTQCh. 6 - Prob. 19CTQCh. 6 - Prob. 20CTQCh. 6 - Prob. 21CTQCh. 6 - Prob. 22CTQCh. 6 - Prob. 23CTQCh. 6 - Draw a constitutional isomer of pentane,...Ch. 6 - How many H’s are lost from the molecular formula...Ch. 6 - How many ifs are lost from the molecular formula...Ch. 6 - Prob. 27CTQCh. 6 - What is the degree of unsaturation for the example...Ch. 6 - Without counting hydrogens, determine which one of...Ch. 6 - Determine the degree of unsaturation (and draw a...Ch. 6 - a model of each molecule shown above: Is the...Ch. 6 - Prob. 32CTQCh. 6 - Prob. 33CTQCh. 6 - Label each double bond E, Z, or neither. (It may...Ch. 6 - Prob. 35CTQCh. 6 - Prob. 36CTQCh. 6 - Indicate the relationship between each pair....Ch. 6 - Prob. 38CTQCh. 6 - Prob. 1ECh. 6 - Prob. 2ECh. 6 - Using your model of butane (CH3CH2CH2CH3) ,...Ch. 6 - Consider the molecule 1-bromo-2-methylbutane. C3...Ch. 6 - Prob. 5ECh. 6 - Prob. 8ECh. 6 - Prob. 9ECh. 6 - Prob. 10ECh. 6 - Prob. 11ECh. 6 - Prob. 12ECh. 6 - Prob. 13ECh. 6 - Prob. 15ECh. 6 - Prob. 16ECh. 6 - Prob. 17ECh. 6 - Prob. 18ECh. 6 - Prob. 19ECh. 6 - Prob. 20ECh. 6 - Prob. 21ECh. 6 - Double bonds do not rotate freely under normal...Ch. 6 - up an example (not appearing in this ChemActivity)...Ch. 6 - Prob. 24ECh. 6 - Prob. 25E
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Indicate which compounds below can have diastereomers and which cannot. ni See Perlodic Table C Items (6 images) (Drag and drop into the appropriate area below) Br Br Br H. F Br OH OH OH Br OH Brltnarrow_forwardIn the box below draw the tetrahedral representation of the R enantiomer of the amino acid valine, CH3CH(CH3)CH(NH2)CO,H. • Use the wedge/hash bond tools to indicate stereochemistry where it exists. • Do not draw the amino acid in zwitterion form. opy aste ChemDoodlearrow_forwardIn the box below draw the tetrahedral representation of the S enantiomer of the amino acid valine, CH3CH(CH3)CH(NH2)CO,H. • Use the wedge/hash bond tools to indicate stereochemistry where it exists. Do not draw the amino acid in zwitterion form. opy aste ChemDoodlearrow_forward
- In the box below draw the tetrahedral representation of the R enantiomer of the amino acid cysteine, HSCH,CH(NH2)CO,H. • Use the wedge/hash bond tools to indicate stereochemistry where it exists. • Do not draw the amino acid in zwitterion form. P opy aste ChemDoodlearrow_forward2. Compare and contrast the biological roles of the following amino acids the following pairs of amino acids. Once you have documented these role state which member of the pair is most important and why (1-2 sentences) Pair #1 Ketogenic, glucogenic or both Prevalence in peptides and proteins Role in biosynthesis Role in homeostasis (e.g. ammonia) Important in which pathways Aspartic acid Glutamic acid Pair #2 Arginine Histidine Ketogenic, glucogenic or both Prevalence in peptides and proteins Role in biosynthesis I Role in homeostasis (e.g. ammonia) Pair #3 Ketogenic, glucogenic or both Prevalence in peptides and proteins Role in biosynthesis Role in homeostasis (e.g. ammonia) Important in which pathways Tyrosine Phenylalanine MacBook Airarrow_forward5. Properties and IMF: Chlorophyll a and Chlorophyll b are almost identical in structure, but they separated during TLC. Report the RFs you recorded for both molecules. What functional group(s) are different between them? Then explain these RF values by comparing the IMF present in each molecule to those present in the mobile or stationary phase and how this shaped their affinities for each phase. Use the terms RF, IMF, polar, nonpolar, affinity, mobile phase and stationary phase.arrow_forward
- Match the following amino acids with their chemical properties H -N-CIC- A -A ✓ B но с D VE ✓ F ✓ G / CH3 CH3 CH B H но -N-C-C- feed C но -N-C-C- H CH₂ D HO -N-CIC- 1 H CH₂ OH E но ||| -N-C-C- II H CH₂ I CH₂ CH₂ CH₂ T NH3 I. Acidic II. Nonpolar III. Polar uncharged IV, Basic + F O HO -N-C-C- H CH₂ NH₂ G но | || -N-C-C- I H CH₂ 1arrow_forwardI did not understand soultion for the question.. The relationship between Affinity and Association & Dissociation Constant. Four proteins (A-D) all bind the same ligand (X), with different affinities. For protein A & B we know that they have a binding site for X with a Kd (dissociation constant) of 10⁻⁵and 10⁻⁸ M, respectively. For protein C and D we know that they have a binding site for ligand X with a Ka (association constant) of 10³and 10⁵M, respectively. Which protein has the highest affinity for ligand ? Explain your reasoning. How do you make them all in same constant so that values can be compared and the one with highest Ka or lowest Kd (Highest affinity in both cases) can be ruled out.arrow_forward(Thanks for the help if you can break down the steps and provide drawings(needed) during the answer since i am trying to see if i have solved this in the right way not copy :)) Draw the heme environment in oxymyoglobin and explain how the geometric data for the attached oxygen molecule may be understood. Fe O (bond length) = 1.809 Å; O-O (bond length ) = 1.246 Å; Fe - O - O (bond angle) = 123 \degarrow_forward
- Ramachandran plots for two amino acids differ significantly from that shown in Figure 2.23. Which two, and why?arrow_forwardWhich question will your experiment investigate? (select one and delete the others) How does temperature affect how lactase drops work to break down lactose into glucose and galactose? How does pH affect how lactase drops work to break down lactose into glucose and galactose? How does agitation (movement) affect how lactase drops work to break down lactose into glucose and galactose? What variable will you change in this experiment? This is also called the Independent variable. How will you change it? What variable will you measure in this experiment to determine the effect of the variable you changed? This is also called the Dependent variable. How will you measure it? What variables will you keep the same in this experiment? Check off the items in this list that you would use for this experiment. You may add any items you feel you would need. O skim milk (which contains lactose) O Lactase drops O Graduated cylinder Eye dropper Ice cubes O pH paper O Hot plate Mixing spoon O…arrow_forwardOrder the following types of bonding from strongest (=1) to weakest (=5) in a typical biochemical system. lonic interactions. Hydrogen bonds. London dispersion interactions. Dipole interactions. Covalent bonds.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning