
Concept explainers
(a)
Interpretation:
The six-carbon backbones to be highlighted that will need a double bond such that it’s a constitutional isomer of molecules in column 2 of Model 7.
Concept Introduction:
The isomer which has the same molecular formula, but different bonding patterns or different connectivity is said to be constitutional isomer.
(b)
Interpretation:
The reason for the 2 structures given below to not form constitutional isomer needs to be determined.
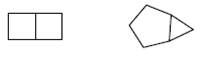
Concept Introduction:
The type of isomers that have the same molecular formula, but their bonding patterns are different, or their connectivity is not the same is said to be constitutional isomer.
(c)
Interpretation:
Whether the given statement is true or false needs to be determined.
Concept Introduction:
An isomer which has the same molecular formula, but the pattern of bonding or connectivity is different is said to be a constitutional isomer.
Want to see the full answer?
Check out a sample textbook solution
Chapter 6 Solutions
Organic Chemistry: A Guided Inquiry
- keferences Draw the structure(s) of the major organic product(s) of the following reaction. H. PPH3 THE • Use the wedge/hash bond tools to indicate stereochemistry where it exists. • If the reaction produces a racemic mixture, draw both stereoisomers. • Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right corner. • Include anionic counter-ions, e.g., I-, in your answer, but draw them in their own sketcher. C opy astearrow_forwardDraw the products of the reaction shown. Electron flow is indicated with curved arrows. H. CH3 ČH3 • Include all valence lone pairs in your answer. • Include counter-ions, e.g., Na*, I', in your submission, but draw them in their own separate sketcher. • Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right corner. • Separate multiple products using the + sign from the drop-down menu. opy ate OFarrow_forward2) For compound 1 shown: a. Draw all the resonance contributors, if any, including proper electron pushing arrows and resonance arrows. A 7-membered ring template is included to get you started." b. Within the box given, clearly draw the resonance hybrid, if any, including partial charges where appropriate. Any ambiguity will be marked wrong. resonance hybridarrow_forward
- 1. Draw and name all the noncyclic constitutional isomers of pentene C5H10. (Without cyclic structures, there are five.). Follow the steps given below. Step 1. The three possible carbon skeletons with 5 carbons and 10 hydrogens in the respective boxes provided below has already been drawn for you. Step 2. Use the different carbon skeletons as a starting point to draw isomers with different double bond locations within each structure. Draw the partially expanded or line structural formulas of the isomers that arise from varying the location of the double bond f in the left column (add the hydrogen atoms for clarification). The number of possible isomers (structural arrangements) of each carbon skeleton is provided. Step 3. Name the IUPAC isomers in the right column. Step 4. Review the IUPAC names to ensure that the first carbon of the longest chain provides the lowest number to the double bond and that all carbons have 4 bonds. IUPAC name of isomers Structural Formula of Isomers A). C…arrow_forwardited Draw both resonance structures of the most stable carbocation intermediate in the reaction shown. od m... • You do not have to consider stereochemistry. . Do not include anionic counter-ions, e.g., I', in your answer. • Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom Separate resonance structures using the → symbol from the drop-down menu. ● CH4 + ? HBrarrow_forwardDraw the products of the reaction shown. Electron flow is indicated with curved arrows. H • Include all valence lone pairs in your answer. • Include counter-ions, e.g., Na+, I", in your submission, but draw them in their own separate sketcher • Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the botton Separate multiple products using the + sign from the drop-down menu. + ? ChemDoodle ▾ {n [arrow_forward
- Draw the products of the reaction shown. Electron flow is indicated with curved arrows. • Include all valence lone pairs in your answer. . Include counter-lons, e.g., Na*, F, in your submission, but draw them in their own separate sketcher. • Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right corner. Separate multiple products using the + sign from the drop-down menu. +98) 24 Y SIF Nextarrow_forward[References] Follow the flow of electrons indicated by the curved arrows in the following polar reaction. Draw all products that result, including any inorganic ions. H-O: CH3 H. H. Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right corner. • Include all valence lone pairs in your answer. P. opy aste [片 ChemDoodle remove Previous Show Hint T***arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
