
Interpretation: To determine assumptions made by Bohr about the motion of electrons.
Concept Introduction: Bohr was the scientist who gave the postulates for the structure of an atom and the position of electrons in a particular atom.
Answer to Problem 28A
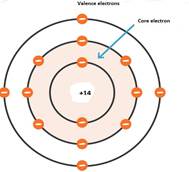
Bohr assumes that electrons would travel in circular paths surrounding the nucleus and do not dissipate energy.
Explanation of Solution
Bohr said that electrons move in the circular path in an atom’s orbital. Proton and neutrons are present in the nucleus with a positive charge. It can be understood by taking the example of the
The postulates of the
- Electrons will travel in a circular path surrounding the nucleus, which will be termed an orbital.
- Every orbital will contain its own definite energy.
- Electrons present in these orbitals do not lose energy.
- The energy level will be indicated by the n, quantum number which will be indicated by n where n is 1,2,3….
Chapter 5 Solutions
Chemistry 2012 Student Edition (hard Cover) Grade 11
- Devise electrochemical cells in which the following reactions could be made to occur. If liquid junctions are necessary, note them in the cell schematic appropriately, but neglect their effects. (a) H2OH + OH¯ (b) 2H2O2 H₂O (c) 2PbSO4 + 2H2O (d) An TMPD PыO₂+ Pb + 4H+ + 20%¯¯ An + TMPD (in acetonitrile, where An and An are anthracene and its anion radical, and TMPD and TMPD are N,N,N',N'-tetramethyl-p-phenylenediamine and its cation radical. Use anthracene potentials for DMF solutions given in Appendix C.3). (e) 2Ce3+ + 2H + BQ 2Ce4+ + H2Q (aqueous, where BQ is p-benzoquinone and H₂Q is p- hydroquinone) (f) Ag +Agl (aqueous) (g) Fe3+ + Fe(CN)6 Fe²+ + Fe(CN) (aqueous)arrow_forwardConsider each of the following electrode-solution interfaces, and write the equation for the elec- trode reaction that occurs first when the potential is moved in (1) a negative direction and (2) a posi- tive direction from the open-circuit potential. Next to each reaction write the approximate potential for the reaction in V vs. SCE (assuming the reaction is reversible). (a) Pt/Cu2+ (0.01 M), Cd2+ (0.01 M), H2SO4(1 M) (b) Pt/Sn2+ (0.01 M), Sn4+ (0.01 M), HCl(1 M) (c) Hg/Cd2+ (0.01 M), Zn2+ (0.01 M), HCl(1 M)arrow_forwardWhat are the major products of both of the organic reactions. Please be sure to use wedge and dash bonds to show the stereochemistry of the products if it is needed. Please include the final product as well as a digram/drawing to show the mechanism of the reaction.arrow_forward
- K Problem 16 of 24 Submit Draw the starting structure that would yield this product under these conditions. Select to Draw 1. NH4Cl, NaCN 2. HCI, H2O, A NH3 + 0arrow_forwardGive detailed me detailed mechanism Solution with explanation needed. Don't give Ai generated solution. avoid handwritten Solutionarrow_forwardShow work with explanation needed. don't give Ai generated solutionarrow_forward
- K Problem 21 of 24 Submit Draw the missing organic structures in the following multistep synthesis. Show the final product at physiological pH (pH = 7.4). Ignore any inorganic byproducts formed. H 0 NH3 Select to Draw HCN H+, H2O Select to Draw Select to Draw Δarrow_forwardShow work with explanation needed. Don't give Ai generated solution. Give correct solutionarrow_forwardK Problem 23 of 24 Submit Draw the product of the reaction shown below at physiological pH (pH = 7.4). Ignore inorganic byproducts. S O 1. NH3, 2. HCN 3. H+, H₂O, A Select to Drawarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





