
Principles of General, Organic, Biological Chemistry
2nd Edition
ISBN: 9780073511191
Author: Janice Gorzynski Smith Dr.
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 3, Problem 3.93AP
(a)
Interpretation Introduction
Interpretation:
The bond angles around the indicated atoms in the compound have to be predicted.
The given compound is,
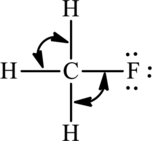
(b)
Interpretation Introduction
Interpretation:
The bond angles around the indicated atoms in the compound have to be predicted.
The given compound is,
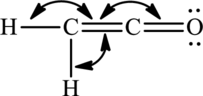
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Indicate the number of single, double, and triple bonds in each of the three compounds.
СH,O has
single bonds,
double bonds, and
triple bonds.
C,H, has
single bonds,
double bonds, and
triple bonds.
Со, has
single bonds,
double bonds, and
triple bonds.
careers
prvacy po ig
tems of use
contact us
belp
2. Infer In a line-angle formula, each line repre-
sents a carbon-carbon bond. Each end of a line, as
well as the intersection of lines, represents a carbon
atom. Knowing that carbon always forms four
covalent bonds, use labeled drawings to summarize
how to determine the number of hydrogen atoms
bonded to each carbon in a line-angle formula.
What are the rules for writing the molecular formula of a simple covalent compound?
Chapter 3 Solutions
Principles of General, Organic, Biological Chemistry
Ch. 3.1 - Predict whether the bonds in the following species...Ch. 3.2 - Write the ion symbol for an atom with the given...Ch. 3.2 - Prob. 3.4PCh. 3.2 - Prob. 3.5PCh. 3.2 - How many electrons and protons are contained in...Ch. 3.2 - Prob. 3.7PCh. 3.3 - Write the formula for the ionic compound formed...Ch. 3.3 - Prob. 3.9PCh. 3.4 - Prob. 3.10PCh. 3.4 - Give the symbol for each ion. a. stannous b....
Ch. 3.4 - Name each ionic compound. a. NaF b. MgO c. SrBr2...Ch. 3.4 - Name each ionic compound. a. CrCl3 b. PbS c. SnF4...Ch. 3.4 - Prob. 3.14PCh. 3.5 - List four physical properties of ionic compounds.Ch. 3.6 - Write the formula for the compound formed when K+...Ch. 3.6 - Prob. 3.17PCh. 3.6 - Name each compound. a. Na2CO3 b. Ca(OH)2 c....Ch. 3.6 - Prob. 3.19PCh. 3.7 - Use electron-dot symbols to show how a hydrogen...Ch. 3.7 - Prob. 3.21PCh. 3.8 - Draw a Lewis structure for each covalent molecule....Ch. 3.8 - Prob. 3.23PCh. 3.8 - Prob. 3.24PCh. 3.9 - Prob. 3.25PCh. 3.9 - Prob. 3.26PCh. 3.10 - Prob. 3.27PCh. 3.11 - Prob. 3.28PCh. 3.11 - Prob. 3.29PCh. 3.11 - Show the direction of the dipole in each bond....Ch. 3.12 - Prob. 3.31PCh. 3.12 - Prob. 3.32PCh. 3 - Which formulas represent ionic compounds and which...Ch. 3 - Which pairs of elements are likely to form ionic...Ch. 3 - Prob. 3.35UKCCh. 3 - Prob. 3.36UKCCh. 3 - Prob. 3.37UKCCh. 3 - Prob. 3.38UKCCh. 3 - Prob. 3.39UKCCh. 3 - Prob. 3.40UKCCh. 3 - Prob. 3.41UKCCh. 3 - Prob. 3.42UKCCh. 3 - Prob. 3.43UKCCh. 3 - Prob. 3.44UKCCh. 3 - Prob. 3.45UKCCh. 3 - Prob. 3.46UKCCh. 3 - (a) Translate each ball-and-stick model to a Lewis...Ch. 3 - Prob. 3.48UKCCh. 3 - Prob. 3.49APCh. 3 - How many protons and electrons are present in each...Ch. 3 - Prob. 3.51APCh. 3 - Prob. 3.52APCh. 3 - Prob. 3.53APCh. 3 - Give the ion symbol for each ion. a. barium ion b....Ch. 3 - Prob. 3.65APCh. 3 - Write the formula for the ionic compound formed...Ch. 3 - Prob. 3.67APCh. 3 - Prob. 3.68APCh. 3 - Name each ionic compound. a. Na2O b. BaS c. PbS2...Ch. 3 - Name each ionic compound. a. KF b. ZnCl2 c. Cu2S...Ch. 3 - Prob. 3.71APCh. 3 - Write formulas to illustrate the difference...Ch. 3 - Prob. 3.73APCh. 3 - Name each ionic compound. a. (NH4)2SO4 b. NaH2PO4...Ch. 3 - Prob. 3.75APCh. 3 - Prob. 3.76APCh. 3 - Prob. 3.77APCh. 3 - Label each statement as true or false. Correct any...Ch. 3 - Prob. 3.79APCh. 3 - Prob. 3.80APCh. 3 - Prob. 3.81APCh. 3 - Prob. 3.82APCh. 3 - Convert the 3-D model of oxalic acid into a Lewis...Ch. 3 - Convert the 3-D model of the general anesthetic...Ch. 3 - Prob. 3.85APCh. 3 - Prob. 3.86APCh. 3 - Prob. 3.87APCh. 3 - Prob. 3.88APCh. 3 - Prob. 3.89APCh. 3 - Prob. 3.90APCh. 3 - Prob. 3.91APCh. 3 - Prob. 3.92APCh. 3 - Prob. 3.93APCh. 3 - Prob. 3.94APCh. 3 - Rank the atoms in each group in order of...Ch. 3 - Prob. 3.96APCh. 3 - Prob. 3.97APCh. 3 - Prob. 3.98APCh. 3 - Prob. 3.99APCh. 3 - Which bond in each pair is more polarthat is, has...Ch. 3 - Prob. 3.101APCh. 3 - Prob. 3.102APCh. 3 - Isobutyl cyanoacrylate is used in medical glues to...Ch. 3 - Prob. 3.104APCh. 3 - Prob. 3.105CPCh. 3 - Prob. 3.106CP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Use the References to access important values if needed for this question. Complete the following structural formula for a neutral molecule by adding H atoms to complete the valence of each atom. Do not introduce any double or triple bonds. C-C-o Then write the molecular formula in the order CHO.arrow_forwardDraw a valid Lewis structure for each species. CH3CH3 CH5N C2H5Brarrow_forwardHow many hydrogens are present around each indicated atom?arrow_forward
- Phosphorus is immediately under nitrogen in the Periodic Table. Predict the molecular formula for phosphine, the compound formed by phosphorus and hydrogen. Predict the H-P-H bond angle in phosphinearrow_forwardUse the Molecular Polarity Simulation to complete the following. Effect of Molecular Geometry on Polarity For each molecule: Sketch each molecule as shown in the simulation. Include arrows to show the bond dipoles as well as a molecular dipole (if present). Circle polar or nonpolar to indicate the polarity of the molecule. Name the molecular geometry (from Part A) H2O vs CO2 H2O CO2 polar nonpolar polar nonpolar Molecular Geometry Molecular Geometry Question 1a: How does the molecular geometry (linear vs bent) affect the molecular polarity?arrow_forwardPart E: Polarity of Molecules After watching the video on Polarity of Molecules, determine if the entire molecule for each structure shown below is polar or nonpolar. In addition, be sure to indicate polar bonds using arrows or partial charge (delta ō signs) as shown in the first example. Remember, a C-H bond is nonpolar and nonpolar bonds would not require an arrow in this process. 3D Sketch of Molecule Polar or Nonpolar? Formula Molecular Geometry Tetrahedral &- CH2Cl2 Polar CH3CI Tetrahedral CI H. CC14 Tetrahedral CI Co Cl CI CI NH3 Trigonal pyramidal H. H. SO2 Bent CO2 Linear 180° 0=C=0 H2CO Trigonal planar H. H.arrow_forward
- Draw an acceptable Lewis structure for each compound, assuming the atoms are connected asarranged. Hydrogen cyanide (HCN) is a poison, formaldehyde (H2CO) is a preservative, and glycolic acid (HOCH2CO2H) is used to make dissolving sutures.arrow_forwardArsenic is a toxic solid often used in the past as a component of pesticides. Counting only s & p electrons, how many valence electrons are present in a neutral atom of arsenic?arrow_forwardHow many hydrogen atoms are present around each highlighted carbon atom in the following molecules? What is the molecular formula for each molecule? Both compounds are active ingredients in some common sunscreens.arrow_forward
- What would be the name for the molecular formula: "P2O3"? which one below phosphorous dioxide dipotassium trioxide diphosphorous trioxide Triphosphorous dioxidearrow_forwardDraw the following molecule and answer the questions below. H3CCHCHC(O)CH2CCH What is the valence electron count? How many total sigma bonds? How many total pi bonds? How many unshared electron pairs (lone pairs)? What is the bond angle of the C(O)C? What is the bond angle of the CCH? What are 3 functional groups contained within the molecule?arrow_forwardChoose the correct pair of formula-name. O HP3 -Phosphorus trihydride O PH3 -Phosphorus trihydride O PH3 -Hydrogen phosphide O P2H3 -Phosphorus hydridearrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co

General, Organic, and Biological Chemistry
Chemistry
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Brooks / Cole / Cengage Learning

Living By Chemistry: First Edition Textbook
Chemistry
ISBN:9781559539418
Author:Angelica Stacy
Publisher:MAC HIGHER

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co
Stoichiometry - Chemistry for Massive Creatures: Crash Course Chemistry #6; Author: Crash Course;https://www.youtube.com/watch?v=UL1jmJaUkaQ;License: Standard YouTube License, CC-BY
Bonding (Ionic, Covalent & Metallic) - GCSE Chemistry; Author: Science Shorts;https://www.youtube.com/watch?v=p9MA6Od-zBA;License: Standard YouTube License, CC-BY
General Chemistry 1A. Lecture 12. Two Theories of Bonding.; Author: UCI Open;https://www.youtube.com/watch?v=dLTlL9Z1bh0;License: CC-BY