
Concept explainers
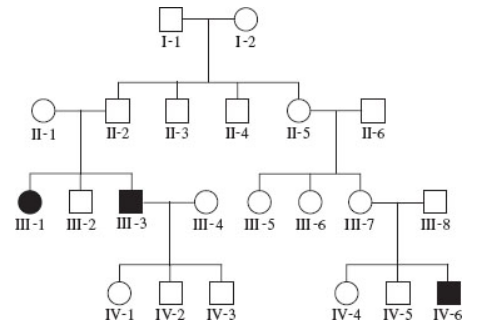
Hurler syndrome is due to a mutation in a gene that encodes a protein called α-l-iduronidase. This protein functions within lysosomes as an enzyme that breaks down mucopolysaccharides (a type of polysaccharide that has many acidic groups attached). When this enzyme is defective, excessive amounts of the mucopolysaccharides dermatan sulfate and heparin sulfate accumulate within the lysosomes, especially in liver cells and connective tissue cells. This accumulation leads to symptoms such as an enlarged liver and spleen, bone abnormalities, corneal clouding, heart problems, and severe neurological problems. The pedigree below contains three members affected with Hurler syndrome, indicated with black symbols. Based on this pedigree, does this syndrome appear to follow autosomal recessive, autosomal dominant, X-linked recessive, or X-linked dominant inheritance? Explain your reasoning.
Want to see the full answer?
Check out a sample textbook solution
Chapter 25 Solutions
Genetics: Analysis and Principles
- How does the loss of the Adenomatous polyposis coli (APC) protein lead to the deregulation of Beta-catenin functions in FAP patients?arrow_forwardNeurofibromatosis type 1 (NF1) is an inherited is an inheritent dominant disorder. The phenotype usually involves the production of many skin neurofibromas. Answer the following questions about the disorder: a) Are the NF1 neurofibromatosis-causing mutations that are inherited by affected children from affected parents likely to be loss-of-function or gain-of-function mutations? b) Neurofibromin, the protein product of NF1, is associated with the Ras protein. Ras is involved in the transduction of extracellular signals from growth factors. The active form of Ras is complexed with GTP; the inactive form is complexed with GDP. Would the wild-type neurofibromin protein favor the formation of Ras-GTP or Ras-GDP? c) Which of the following events in a normal cell from an individual inheriting a neurofibromatosis-causing allele could cause the descendents of that cell to turn into a neurofibroma? i. A second point mutation in…arrow_forwardIn a rare inherited disorder, called Wilson’s disease, excessive amounts of copper accumulate in liver and brain tissue. A prominent symptom of the disease is the deposition of copper in greenishbrown layers surrounding the cornea, called Kayser–Fleischer rings. A defective ATP-dependent protein that transports copper across cell membranes causes Wilson’s disease. Apparently, the copper transport protein is required to incorporate copper into ceruloplasmin and to excrete excess copper. In addition to a diet low in copper, Wilson’s disease is treated with zinc sulfate and the chelating agent penicillamine (p. 148). Describe how these treatments work. [Hint: Metallothionein has a greater affinity for copper than for zinc.]arrow_forward
- In Cystic Fibrosis, what are the normal and mutated protein function?arrow_forwardGiven that a faulty ribosomal protein is the culprit and causes DBA, discuss the possible role of normal ribosomal proteins. Why might bone marrow cells be more susceptible to such a mutation than other cells?arrow_forwardFamilial hypercholesterolemia (FH) is an inherited disease characterized by high blood levels of cholesterol, xanthomas (lipid-laden nodules that develop under the skin near tendons), and early-onset atherosclerosis (the formation of yellowish plaques within arteries). In the milder form of this disease, patients have half the plasma membrane low-density lipoprotein (LDL) receptors needed for cells to bind to and internalize LDL (a plasma lipoprotein particle that transports cholesterol and other lipids to tissues). These individuals have their first heart attacks in young adulthood. In the severe form of FH, in which affected individuals have no functional LDL receptors, heart attacks begin at about age 8, with death occurring a few years later. Based on what you have learned in this chapter, briefly describe the cellular processes that are defective in FH.arrow_forward
- Receptor-mediated endocytosis is a multistep process that allows relatively large structures/molecules to enter cells. Studies of families with familial hypercholesterolemia (FH) have helped to elucidate the process of receptor-mediated endocytosis. The most common type of mutation that leads to this condition affects the LDL receptor cytoplasmic domain, usually a Tyr-to- Cys amino acid change at a single location. Explain how this specific type of mutation in this domain leads to hypercholesterolemia. Give details here, mentioning proteins and interactions affected so it is clear why such a mutation can lead to this health condition. You will need to say what happens here normally and then how this change leads to a problem. Do not just speak in generalities.arrow_forwardA genetic mutation leads to reduced function/loss of function of the V-ATPase on the surface of cellular lysosomes. Explain the role of the V-ATPase on the lysosomal membrane and the possible consequences of the reduced/lost function of this membrane protein.arrow_forwardMembers of the integrin family are the major cell surface receptors for many ECM proteins such as fibronectin. In experimental studies, single integrin heterodimers have been found to assume more than one structural conformation. Describe the relationship between integrin structures and their activities.arrow_forward
- Two missense mutations in the gene that encodes an enzyme called superoxide dismutase cause a form of amyotrophic lateral sclerosis (ALS, or Lou Gehrig disease). This disease causes loss of neurological function over a 5-year period. One mutation alters the amino acid asparagine (Asn) to lysine (Lys). The other changes an isoleucine (Ile) to a threonine (Thr). List the codons involved and describe how single-base mutations can alter the specified amino acids.arrow_forwardFor each of the following situations, provide a plausible explanation for how it could lead to unrestricted cell division.(a) Colon cancer cells often contain mutations in the gene encoding the prostaglandin E2 receptor. PGE2 is a growth factor required for the division of cells in the gastrointestinal tract.(b) Kaposi sarcoma, a common tumor in people with untreated AIDS, is caused by a virus carrying a gene for a protein similar to the chemokine receptors CXCR1 and CXCR2. Chemokines are cell-specific growth factors.(c) Adenovirus, a tumor virus, carries a gene for the protein E1A, which binds to the retinoblastoma protein, pRb. (d) An important feature of many oncogenes and tumor suppressor genes is their celltype specificity. For example, mutations in the PGE2 receptor are not typically found in lung tumors. Explain this observation. (Note that PGE2 acts through a GPCR in the plasma membrane.)arrow_forwardProtein-X has a calcium (Ca²+)-binding regulatory subunit. Upon calcium (Ca²+) binding the protein changes its own conformation resulting in the conformation change of protein-Y. As a result of this calcium (Ca²+)-dependent protein-X mediated conformational change protein-Y exposes the myosin II head binding sites on the skeletal muscle actin filaments. Identify the protein-X and protein-Y from the following choices. O Protein-X: Troponin; Protein-Y: Tropomysosin O Protein-X: Tropomodulin; CapZ O Protein-X: Nebulin; Protein-Y: Titin Protein-X: Tropomysosin; Protein-Y: Tropomodulinarrow_forward
 Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON
Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education,
Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education, Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company
Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co.
Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co. Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education
Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education





