
Concept explainers
Interpretation:
A molecule of
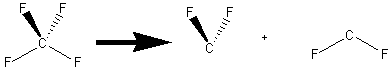
Concept introduction:
A polar covalent bond arises when atoms having different electronegativities are bonded together and the resulting bond dipole points toward the more electronegative atom. Fluorine is more electronegative than carbon. The thick arrows are equal in magnitude and point in opposite directions.
Want to see the full answer?
Check out a sample textbook solution
Chapter 2 Solutions
Organic Chemistry: Principles and Mechanisms (Second Edition)
- Answer Q7, 8, 9 showing detailed explanations.arrow_forwardOne of the orbital interactions we did not consider in this chapter is that between an s AO from one atom and a p AO from another atom in the fashion shown. These orbitals will not interact while in this orientation. Explain why.arrow_forwardI'm very confused on this. please help thank youarrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
