
Organic Chemistry: Principles and Mechanisms (Second Edition)
2nd Edition
ISBN: 9780393663556
Author: Joel Karty
Publisher: W. W. Norton & Company
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 2, Problem 2.5YT
Interpretation Introduction
Interpretation:
In the given structure, the
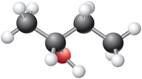
Concept introduction:
A straight line represents a bond that is in the plane of the paper. A wedge signifies a bond, which points toward you and comes out of the plane of the paper. A dash signifies a bond which points away from you and goes behind the plane of the paper. One V is in the plane of the paper whereas the other is perpendicular to the plane of the paper.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Draw the Newman projection of the following compound looking down the bond indicated via the arrow. ( see picture)
please answer this spec question! answer choices are given except toluene and tetrahydrofuran!
Question: How many C-types do each of the following compounds have? Label them a, b,c ... And Circle the carbon type that would be the most downfield. Please answer all 4 parts of the question. Thank you!
Chapter 2 Solutions
Organic Chemistry: Principles and Mechanisms (Second Edition)
Ch. 2 - Prob. 2.1PCh. 2 - Prob. 2.2PCh. 2 - Prob. 2.3PCh. 2 - Prob. 2.4PCh. 2 - Prob. 2.5PCh. 2 - Prob. 2.6PCh. 2 - Prob. 2.7PCh. 2 - Prob. 2.8PCh. 2 - Prob. 2.9PCh. 2 - Prob. 2.10P
Ch. 2 - Prob. 2.11PCh. 2 - Prob. 2.12PCh. 2 - Prob. 2.13PCh. 2 - Prob. 2.14PCh. 2 - Prob. 2.15PCh. 2 - Prob. 2.16PCh. 2 - Prob. 2.17PCh. 2 - Prob. 2.18PCh. 2 - Prob. 2.19PCh. 2 - Prob. 2.20PCh. 2 - Prob. 2.21PCh. 2 - Prob. 2.22PCh. 2 - Prob. 2.23PCh. 2 - Prob. 2.24PCh. 2 - Prob. 2.25PCh. 2 - Prob. 2.26PCh. 2 - Prob. 2.27PCh. 2 - Prob. 2.28PCh. 2 - Prob. 2.29PCh. 2 - Prob. 2.30PCh. 2 - Prob. 2.31PCh. 2 - Prob. 2.32PCh. 2 - Prob. 2.33PCh. 2 - Prob. 2.34PCh. 2 - Prob. 2.35PCh. 2 - Prob. 2.36PCh. 2 - Prob. 2.37PCh. 2 - Prob. 2.38PCh. 2 - Prob. 2.39PCh. 2 - Prob. 2.40PCh. 2 - Prob. 2.41PCh. 2 - Prob. 2.42PCh. 2 - Prob. 2.43PCh. 2 - Prob. 2.44PCh. 2 - Prob. 2.45PCh. 2 - Prob. 2.46PCh. 2 - Prob. 2.47PCh. 2 - Prob. 2.48PCh. 2 - Prob. 2.49PCh. 2 - Prob. 2.50PCh. 2 - Prob. 2.51PCh. 2 - Prob. 2.52PCh. 2 - Prob. 2.53PCh. 2 - Prob. 2.54PCh. 2 - Prob. 2.55PCh. 2 - Prob. 2.56PCh. 2 - Prob. 2.57PCh. 2 - Prob. 2.58PCh. 2 - Prob. 2.59PCh. 2 - Prob. 2.60PCh. 2 - Prob. 2.61PCh. 2 - Prob. 2.62PCh. 2 - Prob. 2.63PCh. 2 - Prob. 2.64PCh. 2 - Prob. 2.65PCh. 2 - Prob. 2.66PCh. 2 - Prob. 2.67PCh. 2 - Prob. 2.68PCh. 2 - Prob. 2.69PCh. 2 - Prob. 2.70PCh. 2 - Prob. 2.71PCh. 2 - Prob. 2.72PCh. 2 - Prob. 2.1YTCh. 2 - Prob. 2.2YTCh. 2 - Prob. 2.3YTCh. 2 - Prob. 2.4YTCh. 2 - Prob. 2.5YTCh. 2 - Prob. 2.6YTCh. 2 - Prob. 2.7YTCh. 2 - Prob. 2.8YTCh. 2 - Prob. 2.9YTCh. 2 - Prob. 2.10YTCh. 2 - Prob. 2.11YTCh. 2 - Prob. 2.12YTCh. 2 - Prob. 2.13YTCh. 2 - Prob. 2.14YTCh. 2 - Prob. 2.15YTCh. 2 - Prob. 2.16YTCh. 2 - Prob. 2.17YTCh. 2 - Prob. 2.18YTCh. 2 - Prob. 2.19YTCh. 2 - Prob. 2.20YT
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Recitation Discussion Problem: USE MOLECULAR MODELS and bring them to recitation. Molecule: 2-methylpentane 1. Draw in bond-line notation. Number all carbons in the pentane backbone. Identify carbons #2 -3. Circle these on your bond-line structure. 2. Draw the Newman projections for all 60° rotations looking down carbons #2-3- starting with the eclipsed conformation. Make sure to keep the front carbon static only rotate the back carbon. Use models. You should draw seven Newman projections in total, 4 eclipsed, 3 staggered. The first and last Newman projections (0° and 360°) are identical. See Karty Chapter 4 for examples. 3. Identify the major interactions occurring in each conformer (which bonds are eclipsed; which have gauche interactions, etc.) and RANK associated energy of eclipsed in terms of size of group. Greater energies are involved for larger eclipsed groups. See Karty Chapter 4 energy diagram examples. 4. Plot these conformations on an Energy Diagram (similar to Karty Figure…arrow_forwardPlease answer this NEATLY, COMPLETELY, and CORRECTLY for an UPVOTE. Write the resonance structure that would result from pushing the electrons as indicated by the curved arrow.arrow_forwardPlease help , will provide helpful ratings for solving all 4 subparts only. Draw the skeletal structures that correspond to the following systematic (IUPAC) names.arrow_forward
- Complete the following for the compound shown below. a) Draw the line structure using the wedge/dash bond convention to show the correct stereochemistry. b) Draw both chair conformations, and indicate that they are in equilibrium using appropriate arrows. c) Label the most stable structure. d) Label the most structure that is the major conformer in solution. e) Explain your answers to c) and d) as we did in class, and Loudon did in the text.arrow_forward[Review Topics] Examine the geometry of the molecule in the 3D window. . Double-click on an atom, and then move cursor over another one to get a distance (click off of the molecule to end) Double-click on an atom, click on a second and move cursor over a third to get the angle about the second atom (click off of the molecule to end) Submit Answer Ⓒ Retry Entire Group JSmol The molecule has a carbonyl group. The molecule has one shorter carbon-to-oxygen bond distance and one longer one. A general functional group representation of the molecule i The substance is a(n) [References] 9 more group attemp RCHO RCOR* RCOOR RCOOH where R and R* are carbon groups. Previous Next Save anarrow_forwardQuestion: A Newman Projection of a tetrahydropyran is drawn below. Draw a bond line-angle representation of the substituted tetrahydropyran in the following figure. Be sure to show realtive positions in the bond-line structure by using either dashes or wedges.arrow_forward
- Answer all otherwise skip it,arrow_forwardDate No. Structural formula Draw the condensed of the compound.arrow_forwardDraw the Newman projection so that it corresponds to the molecule and conformation shown when viewed down the red bond in the direction of the red arrow. Your projection should be oriented as shown by the arrow marked up. So the CH2SH group on the front carbon should be above the H and H3C groups, no matter which template you usearrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning