
(a)
Interpretation:
The given substituted acetic acid has to be prepared using malonic ester synthesis.
Concept Introduction:
Malonic ester is a versatile starting material for the formation of new carbon-carbon bonds. It is because of the presence of acidic α-hydrogens between two carbonyl groups, the nucleophilicity of the enolate anion formed by the loss of α-hydrogen and the ability of the product to undergo decarboxylation after hydrolysis of ester.
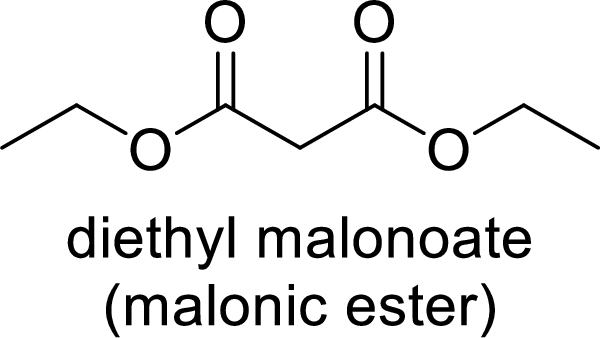
(b)
Interpretation:
The given substituted acetic acid has to be prepared using malonic ester synthesis.
Concept Introduction:
Malonic ester is a versatile starting material for the formation of new carbon-carbon bonds. It is because of the presence of acidic α-hydrogens between two carbonyl groups, the nucleophilicity of the enolate anion formed by the loss of α-hydrogen and the ability of the product to undergo decarboxylation after hydrolysis of ester.
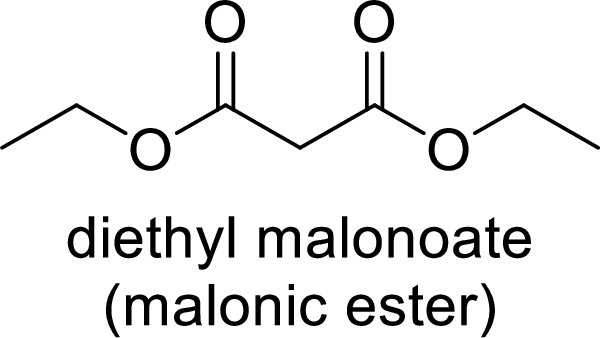
(c)
Interpretation:
The given substituted acetic acid has to be prepared using malonic ester synthesis.
Concept Introduction:
Malonic ester is a versatile starting material for the formation of new carbon-carbon bonds. It is because of the presence of acidic α-hydrogens between two carbonyl groups, the nucleophilicity of the enolate anion formed by the loss of α-hydrogen and the ability of the product to undergo decarboxylation after hydrolysis of ester.
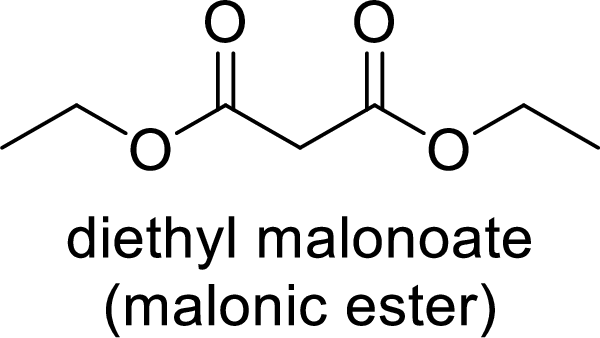
Trending nowThis is a popular solution!

Chapter 19 Solutions
Organic Chemistry
- Show how the following compound can be prepared by a malonic or acetoacetic ester synthesis (or other B-dicarbonyl) reaction:arrow_forwardShow how to synthesize the following compound using either the malonic ester synthesis or the acetoacetic ester synthesis. Q.) 4-Phenyl-2-butanonearrow_forwardShow how the malonic ester synthesis is used to prepare 2-benzylbutanoic acid.Sarrow_forward
- Show how to synthesize the following compound using either the malonic ester synthesis or the acetoacetic ester synthesis. Q.) Cyclobutyl methyl ketonearrow_forwardShow how the following compounds can be made using the malonic ester synthesis.(a) 3-phenylpropanoic acidarrow_forwardShow how the p-chloroaniline acid can be synthesized from benzene:arrow_forward
- Show how the m-hydroxybenzoic acid can be synthesized from benzene:arrow_forwardIf methanol rather than water is added at the end of a Hell–Volhard–Zelinskii reaction, an ester rather than an acid is produced. Show how you would carry out the following transformation, and propose a mechanism for the ester forming step.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

