
(a)
Interpretation:
The way by which alkylation or acylation of an enamine can be used to convert acetophenone into the given compound has to be shown.
Concept introduction:
Enamine:
An enamine is a compound where the lone pair of electrons in the nitrogen atom of
It is formed by the reaction of a
Hydrolysis of enamine: Under acidic conditions, enamine reacts with water to give corresponding ketone and secondary amine.
Here, the bond between nitrogen and the
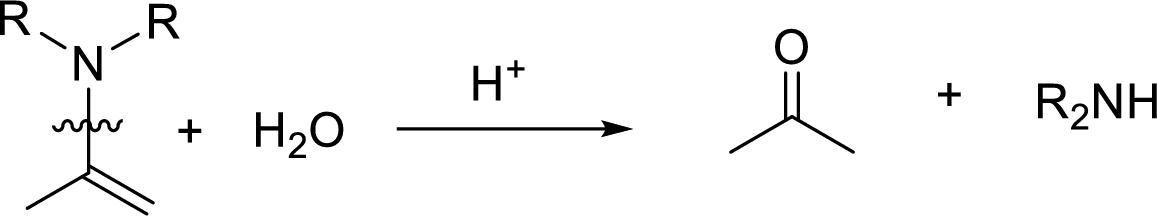
(b)
Concept introduction:
Enamine:
An enamine is a compound where the lone pair of electrons in the nitrogen atom of
It is formed by the reaction of a ketone or aldehyde and a secondary amine under acidic conditions with the elimination of water molecule.
Hydrolysis of enamine: Under acidic conditions, enamine reacts with water to give corresponding ketone and secondary amine.
Here, the bond between nitrogen and the
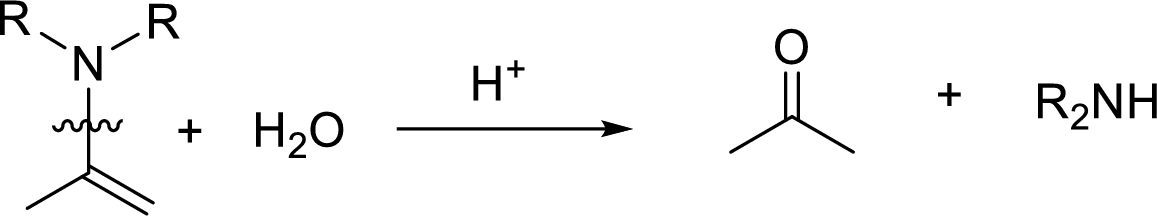
(c)
Concept introduction:
Enamine:
An enamine is a compound where the lone pair of electrons in the nitrogen atom of
It is formed by the reaction of a ketone or aldehyde and a secondary amine under acidic conditions with the elimination of water molecule.
Hydrolysis of enamine: Under acidic conditions, enamine reacts with water to give corresponding ketone and secondary amine.
Here, the bond between nitrogen and the
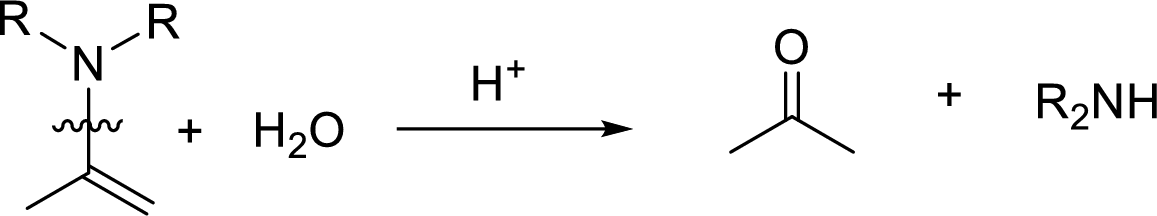
Want to see the full answer?
Check out a sample textbook solution
Chapter 19 Solutions
Organic Chemistry
- Describe how the following compounds could be prepared from cyclohexanone using an enamine intermediate:arrow_forwardAldehydes and ketones react with thiols to yield thioacetals just as they react with alcohols to yield acetals. Predict the product of the following reaction, and propose a mechanism:arrow_forwardHow imines and enamines are converted back to carbonyl compounds by hydrolysis with mild acid ?arrow_forward
- Predict the products formed when cyclohexanone reacts with the following reagents. h) sodium acetylide, then mild H3O+arrow_forwardIndicate how the following compounds can be synthesized from cyclohexanone and any other necessary reagents:arrow_forwardPropose a mechanism for the reaction of benzyl acetate with methylamine. Label theattacking nucleophile and the leaving group, and draw the transition state in which theleaving group leaves.arrow_forward
- How to synthesize 5-nitro-2-hydroxyacetophenone from 2-hydroxyacetophenone?arrow_forwardDraw out the reaction mechanism for cyclohexanol to cyclohexanone. Sodium hypochlorite oxidation of an alcohol to a ketone with the product being cyclohexanone.arrow_forwardProvide the steps to convert the following aldehyde to the given amine product.arrow_forward
- Diazomethane can be used to convert a carboxylic acid to a methyl ester. Propose a mechanism for this reaction.arrow_forwardBisphenol A is made on a large scale by a condensation of phenol with acetone. Suggest an appropriate catalyst, and propose a mechanism for this reaction.arrow_forwardAcetic acid has been mixed with isoamyl alcohol to produce isoamyl acetate giving off a banana smell. Propose a reaction mechanism for this reaction.arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

