
Organic Chemistry
8th Edition
ISBN: 9781305580350
Author: William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 19, Problem 19.70P
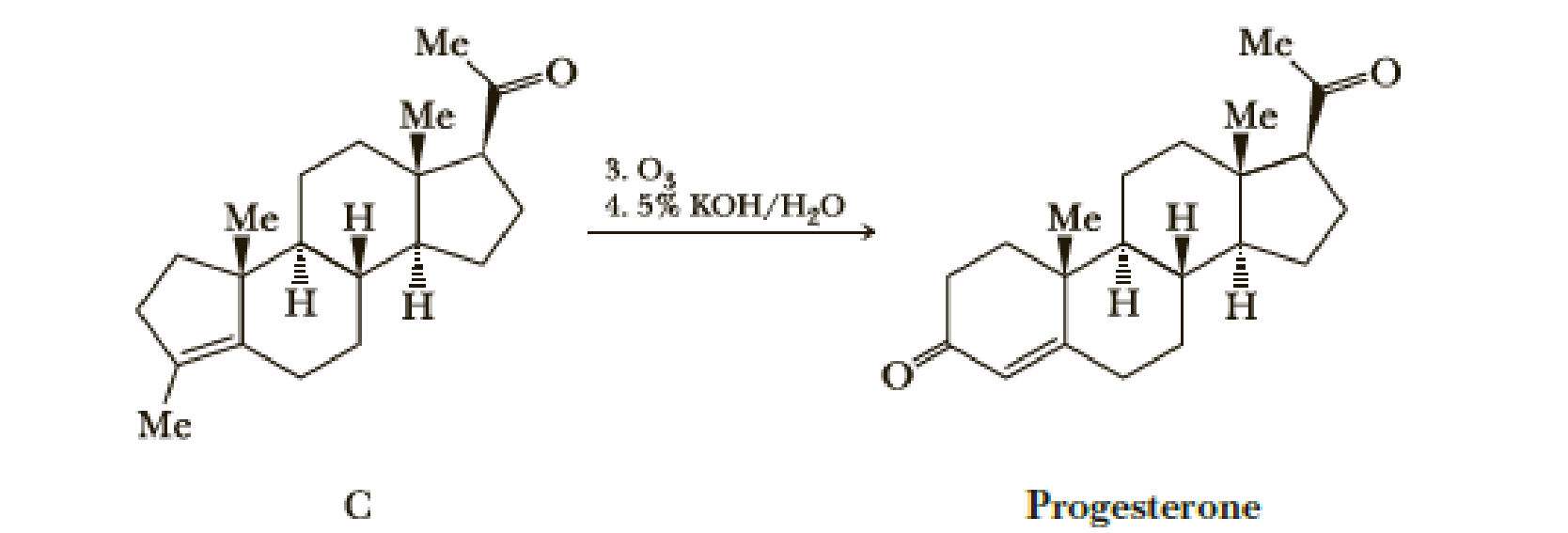
In Problem 7.28, we saw this two-step sequence in Johnson’s synthesis of the steroid hormone progesterone. Propose a structural formula for the intermediate formed in Step 3 and a mechanism for its conversion in Step 4 to progesterone.
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
A step in a synthesis of PGE1 (prostanglandin E1, alprostadil) is reaction of a trisubstituted cyclohexene with bromine to form a bromolactone. Alprostadil is used as temporary therapy for infants born with congenital heart defects that restrict pulmonary blood flow. It brings about dilation of the ductus arteriosus, which in turn increases blood flow in the lungs and blood oxygenation.
Propose a mechanism for formation of This bromolactone, and account for the observed stereochemistry of each substituent on the cyclohexane ring.
Dimethyl disulfide, CH,S–SCH3, found in the vaginal secretions of
female hamsters, acts as a sexual attractant for the male hamster. Write
an equation for its synthesis from methanethiol.
When A is reacted with hot aqueous NaOH, a compound B of molecular formula C8H11NO is produced. With this information, write the correct structure of B and propose the reaction mechanism (step by step, with the correct use of arrows) to obtain B.
Chapter 19 Solutions
Organic Chemistry
Ch. 19.2 - Prob. 19.1PCh. 19.2 - Prob. 19.2PCh. 19.2 - Prob. 19.3PCh. 19.3 - Prob. 19.4PCh. 19.3 - Prob. 19.5PCh. 19.3 - Prob. 19.6PCh. 19.5 - Prob. 19.7PCh. 19.5 - Prob. 19.8PCh. 19.5 - Prob. 19.9PCh. 19.6 - Prob. 19.10P
Ch. 19.6 - Prob. 19.11PCh. 19.7 - Prob. 19.12PCh. 19.8 - Prob. 19.13PCh. 19.8 - Prob. 19.14PCh. 19.8 - Prob. 19.15PCh. 19.8 - Prob. 19.16PCh. 19.9 - Prob. 19.17PCh. 19.9 - Prob. AQCh. 19.9 - Prob. BQCh. 19.9 - Prob. CQCh. 19.9 - Prob. DQCh. 19.9 - Prob. EQCh. 19.9 - Prob. FQCh. 19.9 - Prob. GQCh. 19.9 - Intermediate G in Synthesis III is produced as a...Ch. 19.9 - Prob. IQCh. 19.9 - Prob. JQCh. 19 - Prob. 19.18PCh. 19 - Prob. 19.19PCh. 19 - Prob. 19.20PCh. 19 - Prob. 19.21PCh. 19 - Prob. 19.22PCh. 19 - Prob. 19.23PCh. 19 - Cyclohexene can be converted to...Ch. 19 - Prob. 19.25PCh. 19 - Prob. 19.26PCh. 19 - Prob. 19.27PCh. 19 - Prob. 19.28PCh. 19 - Prob. 19.29PCh. 19 - Prob. 19.30PCh. 19 - Draw structural formulas for the -ketoesters...Ch. 19 - Prob. 19.32PCh. 19 - Prob. 19.33PCh. 19 - Propose a synthesis for each ketone, using as one...Ch. 19 - Prob. 19.35PCh. 19 - Claisen condensation between diethyl phthalate and...Ch. 19 - Prob. 19.37PCh. 19 - Prob. 19.38PCh. 19 - Prob. 19.39PCh. 19 - Enamines normally react with methyl iodide to give...Ch. 19 - Prob. 19.41PCh. 19 - Prob. 19.42PCh. 19 - Prob. 19.43PCh. 19 - Prob. 19.44PCh. 19 - Prob. 19.45PCh. 19 - Prob. 19.46PCh. 19 - Prob. 19.47PCh. 19 - Prob. 19.48PCh. 19 - Prob. 19.49PCh. 19 - Prob. 19.50PCh. 19 - Prob. 19.51PCh. 19 - Prob. 19.52PCh. 19 - Show experimental conditions by which to carry out...Ch. 19 - Prob. 19.55PCh. 19 - The compound 3,5,5-trimethyl-2-cyclohexenone can...Ch. 19 - Prob. 19.57PCh. 19 - Prob. 19.58PCh. 19 - The widely used anticoagulant warfarin (see...Ch. 19 - Following is a retrosynthetic analysis for an...Ch. 19 - Following are the steps in one of the several...Ch. 19 - Prob. 19.62PCh. 19 - Prob. 19.63PCh. 19 - Prob. 19.65PCh. 19 - Prob. 19.67PCh. 19 - Prob. 19.68PCh. 19 - Prob. 19.69PCh. 19 - In Problem 7.28, we saw this two-step sequence in...Ch. 19 - Using your reaction roadmaps as a guide, show how...Ch. 19 - Using your reaction roadmaps as a guide, show how...Ch. 19 - Using your reaction roadmaps as a guide, show how...Ch. 19 - Using your reaction roadmaps as a guide, show how...Ch. 19 - Using your reaction roadmaps as a guide, show how...Ch. 19 - Prob. 19.79PCh. 19 - Prob. 19.80PCh. 19 - Prob. 19.81PCh. 19 - The following molecule undergoes an intramolecular...
Additional Science Textbook Solutions
Find more solutions based on key concepts
The active ingredient in Tylenol and a host of other over-the-counter pain relievers is acetaminophen (C8H9NO2)...
Chemistry: Atoms First
Q1. What is the empirical formula of a compound with the molecular formula
Chemistry: A Molecular Approach (4th Edition)
For Practice 1.1
Is each change physical or chemical? Which kind of property (chemical or physical) is demonst...
Principles of Chemistry: A Molecular Approach (3rd Edition)
4. 38 Strontium has four naturally occurring isotopes, with mass numbers 84, 86, 87, arid 88.
a. Write the atom...
Basic Chemistry (5th Edition)
Practice Problem 1.22 Which of the following alkenes can exist as cis-trans isomers? Write their structures. Bu...
Organic Chemistry
Describe the orbitals used in bonding and the bond angles in the following compounds: a. CH3O b. CO2 c. H2CO d....
Organic Chemistry (8th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- The following sequence of steps converts (R)-2-octanol to (S)-2-octanol. Propose structural formulas for intermediates A and B, specify the configuration of each, and account for the inversion of configuration in this sequence.arrow_forwardTerpin, prepared commercially by the acid-catalyzed hydration of limonene, is used medicinally as an expectorant for coughs. (a) Propose a structural formula for terpin and a mechanism for its formation. (b) How many cis, trans isomers are possible for the structural formula you propose?arrow_forwardClaisen condensation between diethyl phthalate and ethyl acetate followed by saponification, acidification, and decarboxylation forms a diketone, C9H6O2. Propose structural formulas for compounds A and B and the diketone.arrow_forward
- Using your reaction roadmap as a guide, show how to convert 1-butanol and ethanol into racemic 2-ethoxy-1-butanol. You must use 1-butanol and ethanol as the source of all carbon atoms in the ether product. (1) (2) (3) OH (4) 2-ethoxy-1-butanol (racemic) OH OH (c) ethylene oxide Suggest reagents and experimental conditions for each step in this synthesis. From the choices provided, suggest appropriate reagents for each step. More than one reagent may be necessary. Use the minimum number of steps possible. Enter your answer as a letter, or a series of letters, in the order necessary to bring about the steps shown. Reagents: (a) (CH3)3 CO K+, THF (d) H₂SO4, H₂O (b) OsO4, H₂O2 1 Br (e) m-chloroperoxybenzoic acid OH (mCPBA) (f) pyridinium chlorochromate (PCC) + le 2 (9) H₂SO4 (anhydrous) (j) Na, NH3 (1) (h) NaOH (k) NBS, heat (i) PBr3 (N-bromosuccinimide) Previous Nextarrow_forwardUsing your reaction roadmap as a guide, show how to convert 1-butanol and ethanol into racemic 2-ethoxy-1-butanol. You must use 1-butanol and ethanol as the source of all carbon atoms in the ether product. (1) (2) (3) OH (4) 2-ethoxy-1-butanol (racemic) OH OH (c) ethylene oxide Suggest reagents and experimental conditions for each step in this synthesis. From the choices provided, suggest appropriate reagents for each step. More than one reagent may be necessary. Use the minimum number of steps possible. Enter your answer as a letter, or a series of letters, in the order necessary to bring about the steps shown. Reagents: (a) (CH3)3 COK+, THF (d) H₂SO4, H₂O (b) OsO4, H₂O2 Br OH (e) m-chloroperoxybenzoic acid (mCPBA) (f) pyridinium chlorochromate (PCC) + 2 Oly (i) PBr3 (9) H₂SO4 (anhydrous) (h) NaOH (j) Na, NH3 (1) (k) NBS, heat (N-bromosuccinimide) Previous Next Save and Exit ?arrow_forwardSeveral sulfonylureas, a class of compounds containing RSO2NHCONHR, are useful drugs as orally active replacements for injected insulin in patients with adult-onset diabetes. These drugs decrease blood glucose concentrations by stimulating b cells of the pancreas to release insulin and by increasing the sensitivity of insulin receptors in peripheral tissues to insulin stimulation. Tolbutamide is synthesized by the reaction of the sodium salt of p-toluenesulfonamide and ethyl N-butylcarbamate . Propose a mechanism for this step.arrow_forward
- Acetylenic fatty acids are found in many tropical plants. Compound H, C18H2502, is one such compound; it absorbs 5 equivalents of H2 when treated with H2 over a Pd/C catalyst. Ozonolysis of compound H produces the following products: pentanal (CH3(CH2)3CHO) 2-охоасetic acid (HOOCCHO) oxalic acid (HooccOOH) nonanedioic acid (HOOC(CH2)7COOH) Draw the structure of compound H. • You do not have to consider stereochemistry. • You do not have to explicitly draw H atoms. ..arrow_forwardComplete the statements by providing the reagents necessary to complete each step of the synthesis. Ketone reacted with CO₂H and then yielded a derivative, which was treated with 1) (i) C6H5MgBr (ii) H3O+ 2) (i) DIBAL-H (ii). H3O+ 3) Sia2BH,H202, NaOH 4) CH2N2 (diazamethane) CO₂C2H5 5) PCC (an oxidant) 6) H2SO4, H2O, heat 7) HgSO4/ H2SO4 8) C₂H5OH, H3O*, heat TABLE OF REAGENTS to form an unsaturated carboxylic acid, treatment with to yield unsaturated aldehyde as the final product. CHO MacBook Air 9) C6H5 CH2 P(C6H5)3; NaOH 10) PCC (an oxidant) 11) NaCN, H₂SO4 12) PhCH2CHO, H3O*arrow_forwardUsing your reaction roadmap as a guide, show how to convert 1-butanol and ethanol into racemic 2-ethoxy-1-butanol. You must use 1-butanol and ethanol as the source of all carbon atoms in the ether product. (1) (2) (3) OH (4) 2-ethoxy-1-butanol (racemic) OH OH Suggest reagents and experimental conditions for each step in this synthesis. From the choices provided, suggest appropriate reagents for each step. More than one reagent may be necessary. Use the minimum number of steps possible. Enter your answer as a letter, or a series of letters, in the order necessary to bring about the steps shown. Reagents: 1 (a) (CH3)3 CO K+, THF (d) H₂SO4, H₂O (b) OsO4, H₂O2 (c) ethylene oxide Br (e) m-chloroperoxybenzoic acid OH (mCPBA) (f) pyridinium chlorochromate (PCC) || Ⓡ (g) H₂SO4 (anhydrous) (h) NaOH (i) PBr3 (j) Na, NH3 (1) (k) NBS, heat (N-bromosuccinimide) Previous Nextarrow_forward
- Using your reaction roadmap as a guide, show how to convert 1-butanol and ethanol into racemic 2-ethoxy-1-butanol. You must use 1-butanol and ethanol as the source of all carbon atoms in the ether product. (1) (2) (3) OH (4) 2-ethoxy-1-butanol (racemic) OH OH Suggest reagents and experimental conditions for each step in this synthesis. From the choices provided, suggest appropriate reagents for each step. More than one reagent may be necessary. Use the minimum number of steps possible. Enter your answer as a letter, or a series of letters, in the order necessary to bring about the steps shown. Reagents: (c) ethylene oxide (a) (CH3)3 COK, THF (d) H₂SO4, H₂O (b) OsO4, H₂O2 Br OH (e) m-chloroperoxybenzoic acid (mCPBA) (f) pyridinium chlorochromate (PCC) + 2 (9) H₂SO4 (anhydrous) (h) NaOH (i) PBr3 (j) Na, NH3 (1) (k) NBS, heat (N-bromosuccinimide) Previous Nextarrow_forwardIsoerythrogenic acid, C18H26O2, is an acetylic fatty acid that turns a vivid blue on exposure to UV light. On Catalytic hydrogenation over a palladium catalyst, five molar equivalents of hydrogen are absorbed, and stearic acid, CH3(CH2)16CO2H, is produced. Ozonolysis of isoerythrogenic acid yields the following products: formaldehyde, CH2O, malonic acid, HO2CCH2CO2H, adipic acid, HO2C(CH2)4CO2H, and the aldehyde carboxylic acid, OHC(CH2)6CO2H. Provide a structure for isoerythrogenic acid.arrow_forwardSyntheses of each of the following compounds have been reported in the chemical literature. Using the indicated starting material and any necessary organic or inorganic reagents, describe short sequences of reactions that would be appropriate for each transformation. (a) 1,1,5-Trimethylcyclononane from 5,5-dimethylcyclononanonearrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

IR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=_TmevMf-Zgs;License: Standard YouTube License, CC-BY