
Concept explainers
(a)
Interpretation:
To write the mechanism for an uncatalyzed hydrolysis of methyl propionate.
Concept introduction:
The hydrolysis reaction of etser is the reaction in which the breaking of ester bond is done by using water molecule. The product obtained after the ester hydrolysis is the corresponding
The general reaction of an ester to produce in presence of only water is written as,
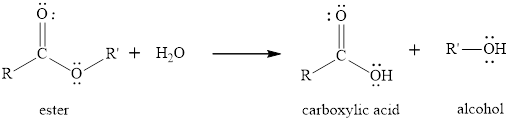
(b)
Interpretation:
To write the mechanism for aminolysis of phenyl formate using methyl amine.
Concept introduction:
The reaction of a carboxylic acid derivative with ammonia, primary and secondary
Want to see the full answer?
Check out a sample textbook solution
Chapter 16 Solutions
Organic Chemistry
- a. Write the mechanism for the following reactions:1. the acid-catalyzed hydrolysis of an imine to a carbonyl compound and a primary amine2. the acid-catalyzed hydrolysis of an enamine to a carbonyl compound and a secondary amine b. How do the two mechanisms differ?arrow_forwardIdentify the reaction type for the following: a. dehydration b. amide synthesis c. esterification d. hydrolysis (in acid) e. hydrolysis (in base) f. hydrationarrow_forward6. Complete the following aqueous reactions: a. N-methylethanamide + NaOH b. N,N-diethylpropanamide + HBr → c. What are these reactions called?arrow_forward
- 12. Murexid test as a group-wide reaction to purine derivatives is based on chemical processes: A. Hydrolytic decomposition B. Electrophilic substitution C. Oxidation D. Nucleophilic additionarrow_forward5. Draw the structure of following compounds: a. methyl butanoate b. propyl ethanoate c. ethyl propanoate d. ethyl 3-methylhexanoate e. butyl hexanoate f. methyl 2-hexyl-3-oxocyclopentanecarboxylatearrow_forwarda. Reaction of acyl compounds with LiAlH4 forms alcohols. b.Reaction of acyl compounds with alcohols forms esters. c.Reaction of carbonyl compounds with LiAlH4 forms alcohols. d.Reaction of carbonyl compounds with alcohols forms hemiacetals. Which are false?arrow_forward
- What two amides are obtained from the reaction of acetyl chloride with an equivalent of ethylamine and an equivalent of propylamine? a. Why is only one amide obtained from the reaction of acetyl chloride with an equivalent of ethylamine and an equivalent of triethylamine?arrow_forwardHow many products are obtained when the phospholipid below is saponified? a. 2 b. 6 c. 5 d. 3arrow_forward12. You can distinguish epinephrine hydrotartrate from norepinephrine hydrotartrate by? A. Water solubility B. Reactions of oxidation with iodine at different pH C. Reactions in general alkaloid precipitation reagents D. Reactions with iron(III) chloride E. Reactions in Fehling's reagent.arrow_forward
- Which could explain the stronger acidity of phenols compared to alcohols. Why? a.pi-electron delocalization b.steric effect c.hydrogen bonding d.hyperconjugationarrow_forwardGive the formula and enumerate the functions of the following carboxylic acids: a. Formic acid b. Citric acid c. Butyric acid d. Pimelic acidarrow_forwardWhat is the name of the product formed as a result of the reaction between benzylamine and ethylacrylate (ethylpropenoate)? A. N-benzylpropenamideB. N-phenylpropanamideC. N-phenylpropenamideD. Ethyl-3-aminobenzylpropanoateE. N-Ethyl-N-benzylpropanamidearrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
