
Concept explainers
(a)
Interpretation:
To identify the reagents used to convert methyl propanoate to the following compounds.
Concept introduction:
A reagent is a substance used to convert one chemical compound into another.
The structural formula for methyl propanoate is:
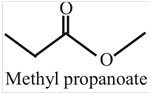
This compound can be converted into different compounds by undergoing a
(b)
Interpretation:
To identify the reagents used to convert methyl propanoate to the following compounds.
Concept introduction:
A reagent is a substance used to convert one chemical compound into another.
The structural formula for methyl propanoate is:

This compound can be converted into different compounds by undergoing a chemical reaction with reagents like water,
(c)
Interpretation:
To identify the reagents used to convert methyl propanoate to the following compounds.
Concept introduction:
A reagent is a substance used to convert one chemical compound into another.
The structural formula for methyl propanoate is:

This compound can be converted into different compounds by undergoing a chemical reaction with reagents like water,
(d)
Interpretation:
To identify the reagents used to convert methyl propanoate to the following compounds.
Concept introduction:
A reagent is a substance used to convert one chemical compound into another.
The structural formula for methyl propanoate is:

This compound can be converted into different compounds by undergoing a chemical reaction with reagents like water,
Want to see the full answer?
Check out a sample textbook solution
Chapter 16 Solutions
Organic Chemistry
- What is the name of the major product formed during the reaction between berzoyl chloride and phenol? a. phenyl benzoate b. benzyl ester C. cyclopentanoate d. benzyl phenoate e. benzenecarboxylic acid O a O b O c earrow_forwardWhich alkyl halides form the carboxylic acids listed here after reaction with sodium cyanide followed by heating the product in an acidic aqueous solution? a. butyric acid b. isovaleric acid c. cyclohexanecarboxylic acidarrow_forwardQUESTION 1 Which of the following compounds has an activating group? A. Benzoic acid B. None in the answer C. Bromobenzene D. Diphenylamine QUESTION 2 Which of the following compounds has a deactivating group attached to the benzene ring? A. Toluene B. Acetanilide C. Phenol D. Salicyclic Acidarrow_forward
- The hydrolysis of an amide in acidic conditions forms A. a carboxylate salt and an alcohol B. a carboxylate salt and an amine C. an alcohol and an amine salt (an ammonium ion) D. a carboxylic acid and an amine salt (an ammonium ion)arrow_forward45. Which of the following compounds has the lowest water solubility? a. butanal b. heptanal c. hexanal d. pentanal 48. Which of the following compounds is the most soluble in water? a. butanal b. heptanal c. hexanal d. pentanal 56. Which of the following compounds will undergo oxidation using potassium dichromate to form a carboxylic acid? A a. A and B only b. A and C only c. B only d. C only H B o Carrow_forwardWhat kind of solvent ingredients is usually used in the concentrations of 4-10 percent in skin care products and their function is to soften skin cells and to lessen wrinkles? A. Ethly acetate B. Alpha hydroxyl acids C. Phenols and phenol derivatives D. Aliphatic alcoholsarrow_forward
- 1. Give the product of this reaction A. Propanal B. Propanol C. Propan-2-olD. Acetic acid E. Propanoic acidarrow_forwardMULTIPLE CHOICE 1. What is the color of the solid produced when acetaldehyde is added with saturated Sodium bisulfite? A. yellow solid B. silver mirror C. brick red precipitate D. white crystalline precipitate 2. An aldehyde that is used in silvering mirrors. * A. propionaldehyde B. formaldehyde C. acetaldehyde D. butyraldehydearrow_forward1. A pleasant smelling liquid having a boiling point of 101°C. What is the structure of this compound? Major IR Absorptions Major lons in the MS 2880-2980 cm-¹ 29 41 1737 (str) 1194 (str) 1166 (str) 56 57 (base) 73 85 101 (small) 116 (molecular ion) 100 1737 500 vavenumber cm-1 157 Transmittance %T ģ 0. liquid film sample 100 80 60 4000 Relative Intensity 9 20 0 10 20 2977 9000 2876 30 4. 40 2000 50 56 60 m/z 70 1500 73 80 1194 85 1166 90 1000 101 repla 100 110 M: 116arrow_forward
- FLAVORANTS: Identify the aldehyde and ketone-containing flavoring compound from each following sources. Choose your best answer from the choices below A. 2-Octanone B. Citral C. Benzaldehyde D. Vanillin E. Cinnamaldehyde F. a-Damascone 1. Вerry 2. Mushroom 3. Lemongrass 4. Cinnamon 5. Almonds Idchyde or ketone formed froarrow_forwardwhich has the greatest acidity? a. 2-hexanol b. valeric acid c. naphtol d. ethyl propionate which produces effervescence when dissolved in 10% sodium bicarbonate? a. 2-hexanol b. valeric acid c. napththol d. ethyl propionatearrow_forwardA chemist wants to make an ester from conversion of alcohol or phenol, what can he used? a. benzamide b. acetic anhydride c. maleic anhydrate d. nonearrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER

