
Concept explainers
Interpretation: The nucleophilic addition reaction occuring steps in the given transamination reaction has to be determined.
Concept introduction:
Amino acids are the molecules containing an
Carbon, hydrogen, oxygen and nitrogen are the key elements in amino acid.
General structure of an amino acid can be drawn as follows,
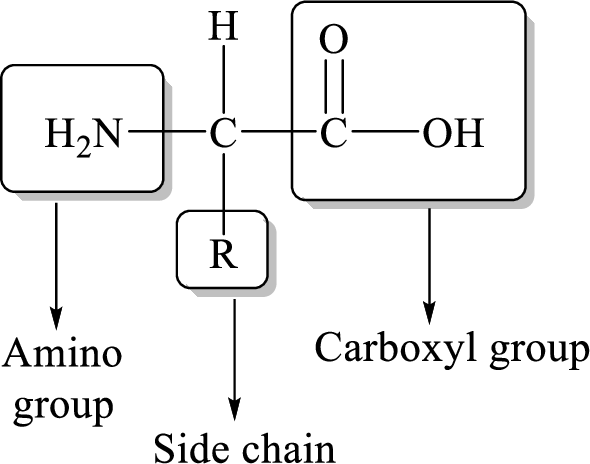
The transfer of an amino group from an amino acid to a ketoacid with the formation of a new amino acid and a new keto acid is known as transamination reaction.
This reaction is catalysed by a group of enzymes called transaminases or aminotransferases. PLP- Pyridoxalphosphate acts as co-factor in this reaction.
Nucleophile: It is negatively charged species which seeks for positive charge and hence donate pair of electrons to positively charged species (electrophiles) which results in the formation of
Nucleophilic addition reaction is an addition reaction where in a chemical compound a
In a nucleophilic addition reaction the first step is the bond formation between a nucleophile and electrophile.
Want to see the full answer?
Check out a sample textbook solution
Chapter 16 Solutions
Organic Chemistry
- What is the intermediate A for the following Stork enamine synthesis? O 1. H₂C. CH3 A 2. H3O* IZ p-TSOH (-H₂O) O CI C12H1402arrow_forwardDraw the structure of the major organic product(s) of the reaction. NaOH H20 Draw the structure of the major organic product(s) of the reaction. OH donon H2O H2SO4 CH3 a. Draw the structure of the tetrahedral intermediate INITIALLY-FORMED in the reaction shown. b. Draw the structures of the organic products of the acyl transfer reaction.arrow_forwardWhether increase or decrease rate of aldol reaction: a. Additional substitution on the ketone b. Electron donating group on the aldehyde c. Electron withdrawing group on the aldehydearrow_forward
- In the following reaction, which chemical species is acting like a nucleophile?arrow_forwardDraw the product of the following reaction sequence. CI 1st attempt Part 1 Draw the product. 1. Mg(s), THF 2. CO₂ (s) 3. H3O+arrow_forwardComplete the following reaction sequence by filling in the intermediate product and the final product. Question 3 CEN H38 Br H3C (conc) Create OscerSketch Answer 3arrow_forward
- 27. What reaction converts the C=O bond to the C=C bond? a. Witten Reaction b. Wittig Reaction c. Winding Reaction d. Whinging Reaction 28. What reagent can reduce imines to primary amines? a. Lithium aluminiumhydride b. Phosphorous pentachloride c. Pyridinium chlorochromate d. Sodium cyanoborohydride 29. What are the steps that can convert imines to amino acids? a. 1. LiAlH₄; Water b. 1. NaCN; 2. Water c. 1. LiAlH₄; 2. Ammonia d. 1. NaCN; 2. Ammonia 30. Cyclohexanone reacts with NaCN to form a.The S-isomer b.The R-isomer c.A racemic mixture d.None of the abovearrow_forwardUse the following choices for questions 36-40: SN1 SN2 E1 E2 E1cb 38-39. Proceeds in a concerted path. 40. A mixture of products is obtained in this reaction.arrow_forwardQuestion 39 Consider the reaction and products. Identify the true statement. Br Br NBS/Light 0°C B is the major product because it has the lower energy Transition State. B is the major product because it is the lower energy product. A is the major product because it is the more stable halide. A is the major product because its pathway has the lower activation energy.arrow_forward
- 1. Complete the following reactions. a. b. C. N HIN 1. LiAlH4 2. H₂O* 1. NaAlH4 2. H₂O* 1. LiAlH4 2. H₂O*arrow_forwardPlace the intermediates in the correct order to complete the mechanism for the following reaction. Reaction: HO H. но, HO H. OH HO. H.arrow_forwardcomplete the reaction 68arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co

