
Organic Chemistry
8th Edition
ISBN: 9781305580350
Author: William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 16.8, Problem BQ
The given mechanism of transamination reaction is shown below,
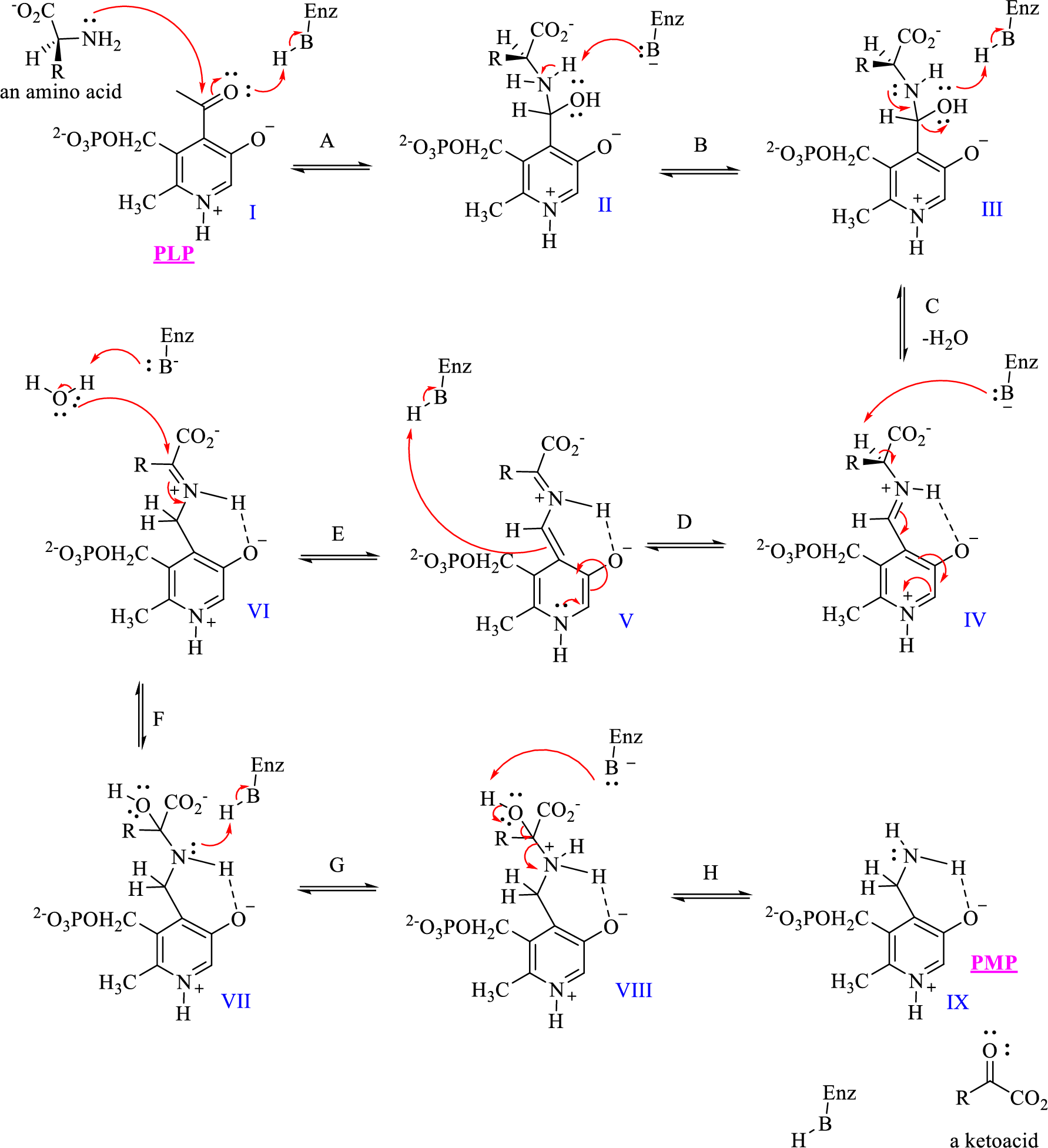
Which structures would be classified as tetrahedral intermediates derived from addition reactions?
- 1. II, III, VII, and VIII
- 2. IV, V, and VI
- 3. II, IV, and VIII
- 4. II and VI
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
What is the intermediate A for the following Stork enamine synthesis?
O
1.
H₂C.
CH3
A
2. H3O*
IZ
p-TSOH
(-H₂O)
O
CI
C12H1402
What is the structure of intermediate A?
1) BH3
2) NaOH, H2O2, H₂O
A
B
Which of these reactions will go through a benzyne intermediate?
CI.
CH.CI
AICI,
NaOH
NaOH
3
CI
170 atm, 360°
H3C.
KMNO,
4
CI
O none of them
01
O all of them
0 2
0 3
04
2.
Chapter 16 Solutions
Organic Chemistry
Ch. 16.1 - Write the IUPAC name for each compound. Specify...Ch. 16.1 - Write structural formulas for all aldehydes with...Ch. 16.1 - Write the IUPAC name for each compound.Ch. 16.5 - Prob. 16.4PCh. 16.6 - Prob. 16.5PCh. 16.7 - Prob. 16.6PCh. 16.7 - Write a mechanism for the acid-catalyzed...Ch. 16.8 - Prob. 16.8PCh. 16.8 - The given mechanism of transamination reaction is...Ch. 16.8 - The given mechanism of transamination reaction is...
Ch. 16.8 - The given mechanism of transamination reaction is...Ch. 16.8 - Prob. DQCh. 16.8 - Prob. EQCh. 16.8 - The given mechanism of transamination reaction is...Ch. 16.9 - Predict the position of the following equilibrium.Ch. 16.9 - Draw a structural formula for the keto form of...Ch. 16.10 - Prob. 16.11PCh. 16.11 - What aldehyde or ketone gives these alcohols upon...Ch. 16.11 - Prob. 16.13PCh. 16 - Prob. 16.14PCh. 16 - Prob. 16.15PCh. 16 - The infrared spectrum of compound A, C6H12O, shows...Ch. 16 - Following are 1H-NMR spectra for compounds B...Ch. 16 - Draw structural formulas for the product formed by...Ch. 16 - Suggest a synthesis for the following alcohols...Ch. 16 - Show how to synthesize the following alcohol using...Ch. 16 - 1-Phenyl-2-butanol is used in perfumery. Show how...Ch. 16 - Prob. 16.22PCh. 16 - Draw structural formulas for (1) the...Ch. 16 - Show how to bring about the following conversions...Ch. 16 - Prob. 16.25PCh. 16 - Wittig reactions with the following -chloroethers...Ch. 16 - Prob. 16.27PCh. 16 - Prob. 16.28PCh. 16 - 5-Hydroxyhexanal forms a six-membered cyclic...Ch. 16 - Prob. 16.30PCh. 16 - Prob. 16.31PCh. 16 - Propose a mechanism to account for the formation...Ch. 16 - Prob. 16.33PCh. 16 - Prob. 16.34PCh. 16 - Show how to bring about the following conversion.Ch. 16 - A primary or secondary alcohol can be protected by...Ch. 16 - Prob. 16.37PCh. 16 - Prob. 16.38PCh. 16 - Prob. 16.39PCh. 16 - Prob. 16.40PCh. 16 - The following molecule belongs to a class of...Ch. 16 - When cis-2-decalone is dissolved in ether...Ch. 16 - Prob. 16.43PCh. 16 - Prob. 16.44PCh. 16 - The following bicyclic ketone has two -carbons and...Ch. 16 - Propose a mechanism for this reaction.Ch. 16 - The base-promoted rearrangement of an -haloketone...Ch. 16 - If the Favorskii rearrangement of...Ch. 16 - (R)-Pulegone, readily available from pennyroyal...Ch. 16 - (R)-Pulegone is converted to (R)-citronellic acid...Ch. 16 - Starting with cyclohexanone, show how to prepare...Ch. 16 - Show how to convert cyclopentanone to these...Ch. 16 - Prob. 16.53PCh. 16 - Prob. 16.54PCh. 16 - Prob. 16.55PCh. 16 - Following is the structural formula of Surfynol, a...Ch. 16 - Prob. 16.57PCh. 16 - Propose a mechanism for this isomerization.Ch. 16 - Starting with acetylene and 1-bromobutane as the...Ch. 16 - Prob. 16.60PCh. 16 - Prob. 16.61PCh. 16 - Prob. 16.62PCh. 16 - Prob. 16.63PCh. 16 - Prob. 16.64PCh. 16 - All rearrangements we have discussed so far have...Ch. 16 - In dilute aqueous base, (R)-glyceraldehyde is...Ch. 16 - Treatment of -D-glucose with methanol in the...Ch. 16 - Treating a Grignard reagent with carbon dioxide...Ch. 16 - Prob. 16.69PCh. 16 - Prob. 16.70PCh. 16 - Prob. 16.71PCh. 16 - Prob. 16.72PCh. 16 - Write the products of the following sequences of...Ch. 16 - Using your reaction roadmaps as a guide, show how...Ch. 16 - Using your reaction roadmaps as a guide, show how...Ch. 16 - Using your reaction roadmaps as a guide, show how...Ch. 16 - Using your reaction roadmaps as a guide, show how...Ch. 16 - Prob. 16.78PCh. 16 - Prob. 16.79PCh. 16 - Prob. 16.80PCh. 16 - Prob. 16.81P
Additional Science Textbook Solutions
Find more solutions based on key concepts
Determine the de Brogue wavelength of a. an electron moving at 1/10 the speed of light. b. a 400 g Frisbee movi...
Inorganic Chemistry
Practice Problem 1.22 Which of the following alkenes can exist as cis-trans isomers? Write their structures. Bu...
Organic Chemistry
Determine [OH], [H+], and the pH of each of the following solutions. a. 1.0 M KCl b. 1.0 M KC2H3O2
Chemistry
Give one example from main group chemistry that illustrates each of the following descriptions: (a) Covalent ne...
General Chemistry: Atoms First
Practice Exercise 1
Which of the following factors determines the size of an atom? a. the volume of the nucleus...
Chemistry: The Central Science (14th Edition)
Q2. Which statement best defines chemistry?
a. The science that studies solvents, drugs, and insecticides
b. Th...
Introductory Chemistry (6th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Suppose the ethyl acetoacetate was replaced with the compound below, what will be the effect on the rate of reaction? H3C CH3 F Increase. The fluoride will make the intermediate more stable. Increase. The fluoride will make the intermediate less stable. Decrease. The fluoride will make the intermediate more stable. Decrease. The fluoride will make the intermediate less stable.arrow_forward13*32 Black-and-white photography is a commercial process that involves a phenol. Black-and-white film is coated with an emulsion containing silver bromide or silver iodide crystals that become activated by exposure to light. The activated silver ions then react with hydroquinone in the developing stage as shown in the following balanced equation. All silver halide not activated by light is removed in the fixing process, and the result is a black image (a negative) left by the deposited metallic silver where the film had been struck by fight. In this redox reaction state: What is reduced and what is the reducing agent? What is oxidized and what is the oxidizing agent?arrow_forwardAn elevated level of the enzyme alkaline phosphatase (ALP) in human serum is an indication of possible liver or bone disorder. The level of serum ALP is so low that it is very difficult to measure directly. However. ALP catalyzes a number of reactions, and its relative concentration can be determined by measuring the rate of one of these reactions under controlled conditions. One such reaction is the conversion of p-nitrophenyI phosphate (PNPP) to p-nitrophenoxide ion (PNP) and phosphate ion. Control of temperature during the test is very important; the rate of the reaction increases 1.47 times if the temperature changes from 30 C to 37 C. What is the activation energy for the ALP-catalyzed conversion of PNPP to PNP and phosphate?arrow_forward
- The given mechanism of transamination reaction is shown below, Which structures are imine or iminium intermediates? 1. I, III, and V 2. VII and VIII 3. IV and VII 4. IV, V, and VIarrow_forward5. (Chapter 6) What type of instability will need to be addressed in the intermediate for the following combination of reactants? pi bond with an electrophilic hydrogen 6. (Chapters 6, 8, 9, and 10) Which is more stable and why? CH3 CH3 H3C- c=CH• -CH2 H2C=ċ- -CH-CH3 + 7. (Chapters 6, 7, 8, and 9) Circle the best place to develop a carbanion in the following molecule: CH3 HC -ċ=CH2 8. (Chapters 8, 9, 10, and 11) Write all of the resonance contributors for the following. Label the contributors as major, minor, or contribute equally. CH3 CHarrow_forward5 Aspirin ◆ What is the role of phosphoric acid in the synthesis of acetylsalicylic acid? (Use equations to explain this purpose and present the reaction mechanism).arrow_forward
- Which of the following reaction is mot likely to activate a drug molecule from orally administered inactive parent molecule? a Aromatic hydroxylation b Benzylic carbon oxidation c Hydrolysis d Azo reductionarrow_forwardCompound A is converted to compound B via a one-step reaction with an activation energy of +3 kcal/mol. The energy difference between Compound B and the transition state of the reaction is 7 kcal/mol. What is the AH° for the reaction? O -10 kcal/mol -4 kcal/mol +10 kcal/mol -3 kcal/mol +3 kcal/mol +4 kcal/molarrow_forwardChoose the most appropriate reagent(s) for the conversion of the ketone to the primary amine intermediate. OH NH2 Na Cr207 HSO4 HO reagent (s) N(CH3 H2, Pd B) NABH4, CH3CH,OH NANH2, NH3 1. LIAIH, 2. H, H20arrow_forward
- 70. Determine the sole product of the following second order elimination reaction: NAOET, ETOH 400 K B D.arrow_forward52. In a concerted or single step reaction, A. the rate of forward reaction is equal to the rate of backward reaction. B. the free energy of the reaction is negative C. the free energy of the reaction is positive D. the stability of the transition state controls the rate of the reaction.arrow_forward7) Epoxidation CH,CO,H CH,COH mCPBA mCPBAarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

 Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning



Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:9781938168390
Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:OpenStax

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning
DIGESTER-35 | VITAMINS AND THEIR RELATED COENZYMES| GPAT | NIPER | PHARMACIST| DI; Author: GPAT DISCUSSION CENTER;https://www.youtube.com/watch?v=CGrdNYmho0s;License: Standard YouTube License, CC-BY