
Concept explainers
(a)
Interpretation:
The structural formula for the hemiacetal and then acetal formed in the given reaction has to be drawn.
Concept Introduction:
The structural formula of a compound shows how the atoms are arranged in a molecule and, in particular, shows which
Hydrocarbon compound which contain a hydroxyl group and an alkoxy group is known as Hemiacetals. Hemiacetal can be prepared by the addition of alcohol to an
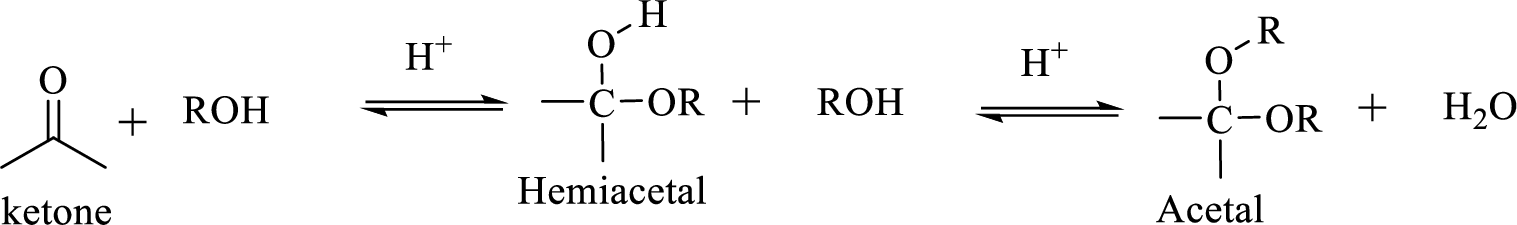
(b)
Interpretation:
The structural formula for the hemiacetal and then acetal formed in the given reaction has to be drawn.
Concept Introduction:
The structural formula of a compound shows how the atoms are arranged in a molecule and, in particular, shows which functional groups are present.
Hydrocarbon compound which contain a hydroxyl group and an alkoxy group is known as Hemiacetals. Hemiacetal can be prepared by the addition of alcohol to an aldehyde or ketone in the presence of an acid catalyst. Hemiacetals react with a second alcohol molecule to produce an acetal, which contains a carbon with two alkoxy groups –OR.
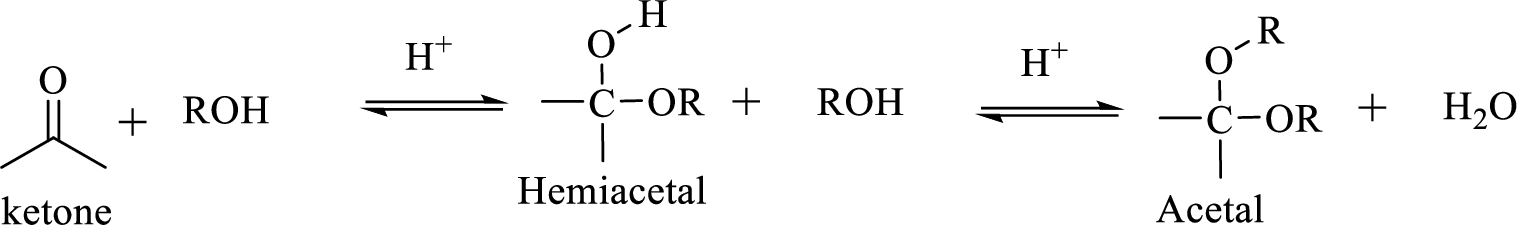
(c)
Interpretation:
The structural formula for the hemiacetal and then acetal formed in the given reaction has to be drawn.
Concept Introduction:
The structural formula of a compound shows how the atoms are arranged in a molecule and, in particular, shows which functional groups are present.
Hydrocarbon compound which contain a hydroxyl group and an alkoxy group is known as Hemiacetals. Hemiacetal can be prepared by the addition of alcohol to an aldehyde or ketone in the presence of an acid catalyst. Hemiacetals react with a second alcohol molecule to produce an acetal, which contains a carbon with two alkoxy groups –OR.
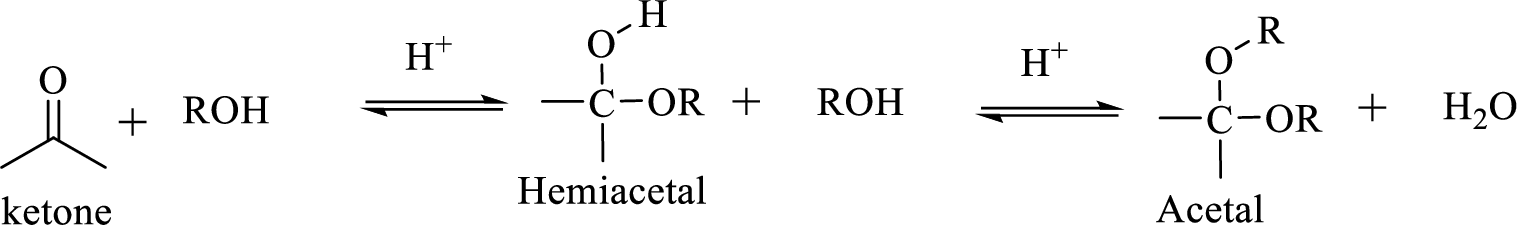
Trending nowThis is a popular solution!

Chapter 16 Solutions
Organic Chemistry
- List the following compounds in order of increasing water solubility: a.ethoxyethane b.propanoic acid c.pentane d.1 butanolarrow_forwardWhat is the role of the acetic acid in the oxidation of Cyclohexanol to Cyclohexanone? Write the balanced chemical reaction between acetic acid and sodium hypochlorite.arrow_forwardDrawing an Enol and a Ketone Formed by Hydration of an Alkyne Draw the enol intermediate and the ketone product formed in the following reaction.arrow_forward
- 1. What is the role of the acetic acid in the oxidation of Cyclohexanol to Cyclohexanone? Write the balanced chemical reaction between acetic acid and sodium hypochlorite.2. How do you neutralize the acetic acid regenerated in the reaction? Write the balanced chemical reaction.arrow_forwardSaponification product of butylpropanoate is: Propanol and Sodium butanoate Butanol and sodium propanoate Butanol and Propanoic acid Butanoic acid and Propanoic acidarrow_forwardEthyl butyrate, CH3CH2CH2CO2CH2CH3, is an artificial fruit flavor commonly used in the food industry for such flavors as orange and pineapple. Its fragrance and taste are often associated with fresh orange juice, and thus it is most commonly used as orange flavoring.It can be produced by the reaction of butanoic acid with ethanol in the presence of an acid catalyst (H+): CH3CH2CH2CO2H(l)+CH2CH3OH(l)H+⟶CH3CH2CH2CO2CH2CH3(l)+H2O(l) a) Given 7.70 g of butanoic acid and excess ethanol, how many grams of ethyl butyrate would be synthesized, assuming a complete 100% yield? b) A chemist ran the reaction and obtained 5.25 g of ethyl butyrate. What was the percent yield? c) The chemist discovers a more efficient catalyst that can produce ethyl butyrate with a 78.0% yield. How many grams would be produced from 7.70 g of butanoic acid and excess ethanol?arrow_forward
- Ethyl butyrate, CH3CH2CH2CO2CH2CH3, is an artificial fruit flavor commonly used in the food industry for such flavors as orange and pineapple. Its fragrance and taste are often associated with fresh orange juice, and thus it is most commonly used as orange flavoring.It can be produced by the reaction of butanoic acid with ethanol in the presence of an acid catalyst (H+): CH3CH2CH2CO2H(l)+CH2CH3OH(l)H+⟶CH3CH2CH2CO2CH2CH3(l)+H2O(l). The chemist discovers a more efficient catalyst that can produce ethyl butyrate with a 78.0% yield. How many grams would be produced from 8.50 gof butanoic acid and excess ethanol? Express your answer in grams to three significant figures.arrow_forwardEthyl butyrate, CH3CH2CH2CO2CH2CH3, is an artificial fruit flavor commonly used in the food industry for such flavors as orange and pineapple. Its fragrance and taste are often associated with fresh orange juice, and thus it is most commonly used as orange flavoring.It can be produced by the reaction of butanoic acid with ethanol in the presence of an acid catalyst (H+): CH3CH2CH2CO2H(l)+CH2CH3OH(l)H+⟶CH3CH2CH2CO2CH2CH3(l)+H2O(l) Given 8.50 g of butanoic acid and excess ethanol, how many grams of ethyl butyrate would be synthesized, assuming a complete 100%yield? Express your answer in grams to three significant figures.arrow_forwardDraw the condensed structural formula or skeletal formula of the hemiacetal and acetal products formed by adding CH3CH2OH (ethanol) to butanal.arrow_forward
- Determine the product for octanal treated with an oxidizing agent. Group of answer choices octanoic acid octyloctanoate octanone octanolarrow_forwardDescribe the chemical properties of acid chlorides.arrow_forwardDraw the pyrrole that would form in each of the following reactions. a) b) COOEt NH3 Ph cat HCI, heat NH2 cat HCI, heat c) 2 equiv NH2arrow_forward
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning




