
(a)
Interpretation:
General structural formula for ester has to be written.
Concept Introduction:
Carbonyl groups are the one which contain a double bond between carbon and oxygen atom.
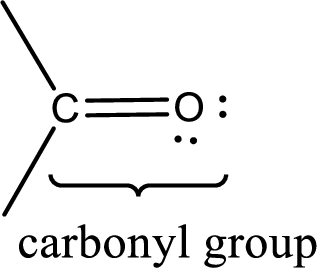
If a hydroxyl group is attached to a carbonyl group means it is known as carboxyl group. This can be represented as shown below,
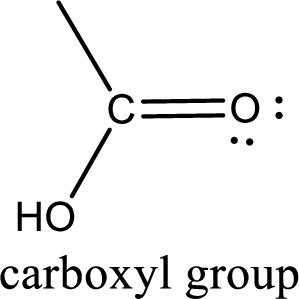
(b)
Interpretation:
General structural formula for acid chloride has to be written.
Concept Introduction:
Carbonyl groups are the one which contain a double bond between carbon and oxygen atom. Aldehydes and ketones possess this carbonyl functional group in it. The structural representation of a carbonyl group can be given as shown below,
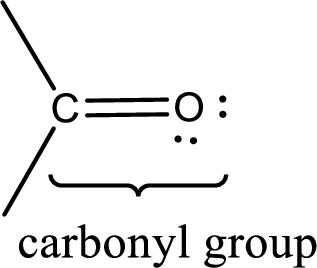
If a hydroxyl group is attached to a carbonyl group means it is known as carboxyl group. This can be represented as shown below,
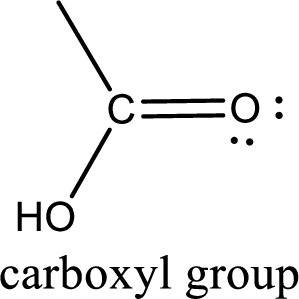
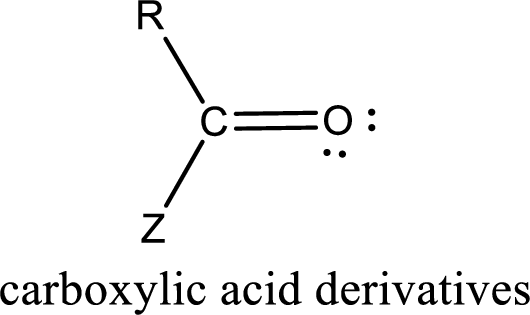
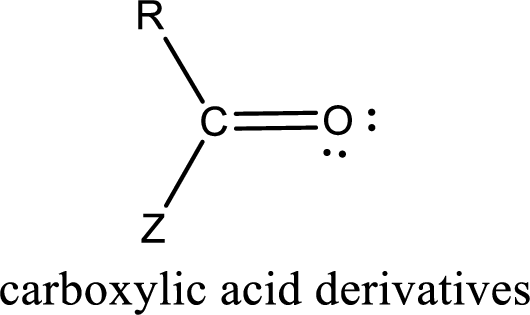
Carboxylic acid derivatives are the ones that are synthesized from or converted to a carboxylic acid. The generalized structural representation of carboxylic acid derivatives is shown below,
(c)
Interpretation:
General structural formula for amide has to be written.
Concept Introduction:
Carbonyl groups are the one which contain a double bond between carbon and oxygen atom. Aldehydes and ketones possess this carbonyl functional group in it. The structural representation of a carbonyl group can be given as shown below,
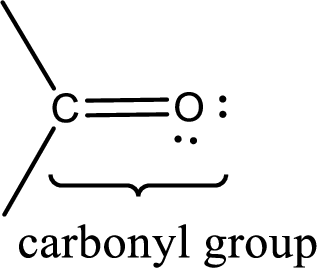
If a hydroxyl group is attached to a carbonyl group means it is known as carboxyl group. This can be represented as shown below,
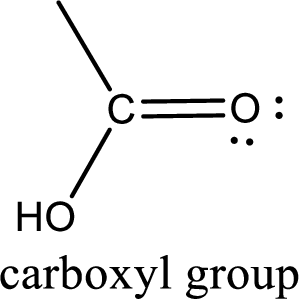
Carboxylic acid derivatives are the ones that are synthesized from or converted to a carboxylic acid. The generalized structural representation of carboxylic acid derivatives is shown below,
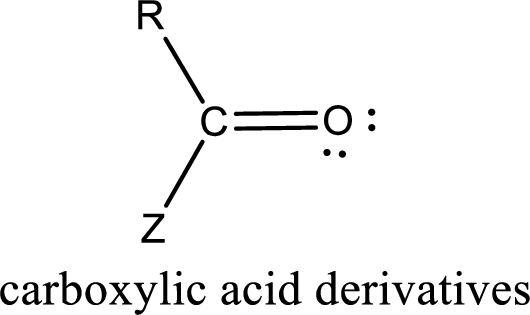
(d)
Interpretation:
General structural formula for acid anhydride has to be written.
Concept Introduction:
Carbonyl groups are the one which contain a double bond between carbon and oxygen atom. Aldehydes and ketones possess this carbonyl functional group in it. The structural representation of a carbonyl group can be given as shown below,
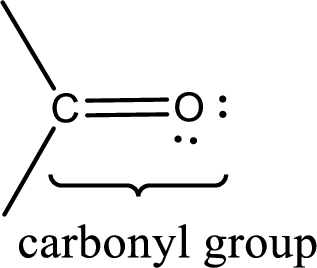
If a hydroxyl group is attached to a carbonyl group means it is known as carboxyl group. This can be represented as shown below,
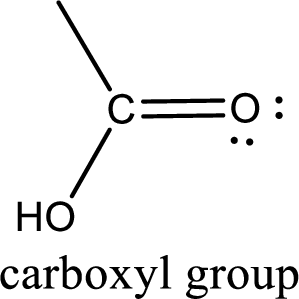
Carboxylic acid derivatives are the ones that are synthesized from or converted to a carboxylic acid. The generalized structural representation of carboxylic acid derivatives is shown below,
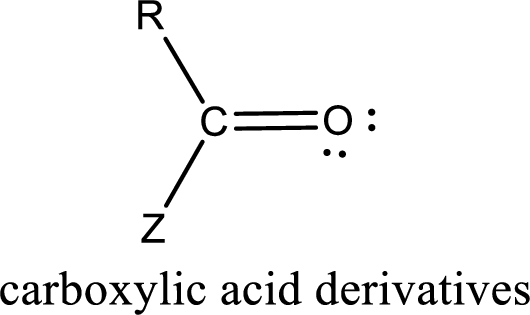
Want to see the full answer?
Check out a sample textbook solution
Chapter 16 Solutions
General, Organic, and Biological Chemistry
- Describe the Properties of Carboxylic Acids and Its Derivatives?arrow_forwardDefine physical properties of carboxylic acid derivatives ?arrow_forwardOdour given when butyric acid react with isoamyl. Odour given when acetic acid react with isoamyl. Odour given when salicylic acid reacts with methanol Odour given when ethanol acid react with cinnamicarrow_forward
- Complete the table provided below for nomenclature of Carboxylic Acid & Derivatives.arrow_forward1. Chief organic component of vinegar a. acetic acid b. formic acid c. benzoic acid d. butanoic acid 2. This term means without water. a. carbonyl b. hydroxyl c. anhydride d. carboyl 3. Compounds containing the cyano group. a. nitriles b. amides c. amines d. nitrates 4. General formula of a Grignard reagent. a. RCOX b. RCN c. RCOOH d. RMgX 5. Organic derivatives of ammonia, derived from replacing one, two or all three hydrogens of the ammonia. a. amide b. amine c. cyan d. nitro 6. Sulfur analogs of alcohols where the O in R-OH is replaced by sulfur. a. Thioesters b. Thiols c. Thioaldehydes d. Thioethers 7. General formula of alkanes. a. CnH2n b. CnH2n+2 c. CnH2n-2 d. R-OH 8. General formula of alkenes. a. CnH2n b. CnH2n+2 c. CnH2n-2 d. R-OH 9. General formula of alkynes. a. CnH2n b. CnH2n+2 c. CnH2n-2 d. R-OH 10. Which is soluble in water? a. methanol b. ethanol c. propanol d. all of the above 11. Which substance will have the highest boiling point? a. methanol b.…arrow_forwardBriefly explain why alcoholic solutions (alcoholic NH2OHꞏHCl, alcoholic KOH, alcoholic HCl) are used in the hydroxamic acid test for esters, what happens if we use other solutions?arrow_forward
- Each of the following acids contains an additional type of functional group besides the carboxyl group. For each acid, specify the non-carboxyl functional group present. 12. Glycolic acid13. Fumaric acid14. Pyruvic acid15. Malic acid16. Tartaric acidarrow_forwardAmide hydrolysis in basic conditions forms A. a carboxylic acid and an amine B. a carboxylate salt and an amine 3. an ester and an amine 4. a carboxylic acid and an amine saltarrow_forwardgive an example of an esterification reaction using hexanoic acid that will result in an esterarrow_forward
- Acid hydrolysis of ------------ produces carboxylic acids. a. Cyanides b. Alcohol c. Ester d. Hydrocarbonarrow_forward1. Which is more reactive? a. carboxylic acid b. acid anhydride c. acid halide d. amides e. esters 2. Removal of water? a. carboxylic acid b. acid anhydride c. acid halide d. amides e. esters 3. Condensation of alcohol and RCOOH? a. carboxylic acid b. acid anhydride c. acid halide d. amides e. estersarrow_forwardCompare and contrast acid hydrolysis and base hydrolysis of carboxylic acid derivatives.arrow_forward
 Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning



