
Interpretation : The relationship between molarity and freezing point depression is to be outlined.
Concept Introduction : Molarity mainly explains the concentration of a solute and the freezing point depression is a colligative property that depends on the number of solute particles and it increases with an increase in the concentration of solute Molarity is the ratio of moles of a solute to the volume of solution in liter. Molarity is the ratio of moles of a solute to the volume of solution in a liter. It is denoted by
The expression for molarity is as follows:
Answer to Problem 12STP
The Freezing point depression is a colligative property that depends on the number of solute particles and it increases with an increase in the concentration of solute as the more the solute more it will alter the freezing point of the solvent or solution.
Explanation of Solution
Molarity mainly explains the concentration of a solute and the freezing point depression is a colligative property that depends on the number of solute particles it increases with an increase in the concentration of solute as the more the solute more it will alter the freezing point of the solvent or solution. Also from the graph, it is clear that freezing point depression increases with an increase in molarity.
The data is given as:
| Molarity | Freezing point depression | ||
The required graph is plotted as:
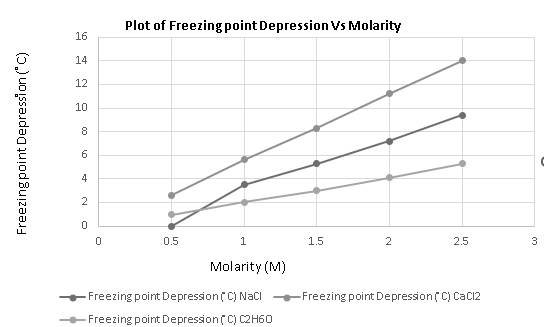
Hence, the freezing point depression increases with an increase in molarity.
The freezing point depression of a solute increases with an increase in molarity.
Chapter 16 Solutions
Chemistry 2012 Student Edition (hard Cover) Grade 11
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





