
Concept explainers
(a)
Interpretation:
To predict the compound formed by the reaction of phosgene with given compound.
Concept introduction:
Carbonyl carbon of phosgene is highly polar in nature because of the presence of electronegative groups attached to it. So phosgene will undergo nucleophilic addition-eliminationreaction. Tetrahedral intermediate is formed when nucleophile is added to the carbonyl carbon of phosgene. The tetrahedral intermediate is unstable therefore chloride ion gets eliminated. The acyl chloride reacts with neutral nucleophile by the following mechanism:
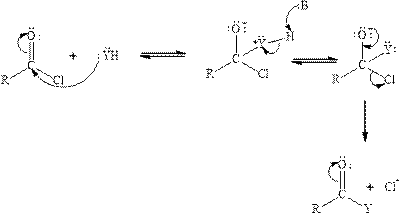
(b)
Interpretation:
To predict the compound formed by the reaction of phosgene with given compound.
Concept introduction:
Carbonyl carbon of phosgene is highly polar in nature because of the presence of electronegative groups attached to it. So phosgene will undergo nucleophilic addition-eliminationreaction. Tetrahedral intermediate is formed when nucleophile is added to the carbonyl carbon of phosgene. The tetrahedral intermediate is unstable therefore chloride ion gets eliminated. The acyl chloride reacts with neutral nucleophile by the following mechanism:

(c)
Interpretation:
To predict the compound formed by the reaction of phosgene with given compound.
Concept introduction:
Carbonyl carbon of phosgene is highly polar in nature because of the presence of electronegative groups attached to it. So phosgene will undergo nucleophilic addition-eliminationreaction. Tetrahedral intermediate is formed when nucleophile is added to the carbonyl carbon of phosgene. The tetrahedral intermediate is unstable therefore chloride ion gets eliminated. The acyl chloride reacts with neutral nucleophile by the following mechanism:

(d)
Interpretation:
To predict the compound formed by the reaction of phosgene with given compound.
Concept introduction:
Carbonyl carbon of phosgene is highly polar in nature because of the presence of electronegative groups attached to it. So phosgene will undergo nucleophilic addition-eliminationreaction. Tetrahedral intermediate is formed when nucleophile is added to the carbonyl carbon of phosgene. The tetrahedral intermediate is unstable therefore chloride ion gets eliminated. The acyl chloride reacts with neutral nucleophile by the following mechanism:

Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
Organic Chemistry (8th Edition)
- What is the major organic product obtained from the following reaction? A. 3-hydroxypropanoic acid B. 3-oxopropanoic acid C. propanedioic acid D. 1,3-propanediolarrow_forwardWhat is the product of the following reaction? a. b. c. d. مر Br 1)LDA, -780 2)EtBrarrow_forwardWhat are the best starting material and reagents needed to make N-methylpropanamide? a. propanol and H2CrO7 b. propanoic acid, methylamine c. propanoic acid, methylamine, and high heat d. propene, H2SO4, H2O, and high heat e. methanoic acid, propylamine, and high heatarrow_forward
- What is the product of the following reaction? a. an alkyl benzene b. an adehyde c. a carboxylic acid d. a ketonearrow_forwardFour test tubes containing different hydrocarbons: acetone, benzoic acid, cyclohexane, and ethylamine were tested for solubility with different solvents: H2O, dilute HCl solution, and a strong solution of NaOH. The results obtained from the solubility test were tabulated below. Determine the content of each test tube A, B, C, and D. Choices: A. Cyclohexane B. Ethylamine C. Benzoic Acid D. Acetonearrow_forward1. Which of the following is NOT a true statement? A. Tartaric acid is present in grapes B. Acetic acid is present in sour milk C. Citric acid is a tricarboxylic acid D. Formic acid is present in insect bites 2. The greater acidity of carboxylic acids compared to alcohols arises primarily from: A. the electron-withdrawing effect of the carboxyl oxygen B. the electron-donating effect of the hydroxyl group C. the acidity of a hydrogens of carboxylic acids D. the resonance stability associated with the carboxylate ion 3. Amides are commonly prepared by reacting acyl halides, esters, or anhydrides with? A. Tertiary amines B. Ammonium hydroxide Please answer all thank you C. Ammonium chloride D. Ammoniaarrow_forward
- What reagent serves as the source of ammonia to deprotonate oxalic acid in the production of calcium oxalate monohydrate? a. ammonium oxalate b. hydrochloric acid c. urea d. ammonium hydroxidearrow_forwardWhat kind of solvent ingredients is usually used in the concentrations of 4-10 percent in skin care products and their function is to soften skin cells and to lessen wrinkles? A. Ethly acetate B. Alpha hydroxyl acids C. Phenols and phenol derivatives D. Aliphatic alcoholsarrow_forwardWhat explains why many aldehydes and ketones can undergo self- condensation reactions in basic conditions? A. The alpha carbon can lose a proton and act like a nucleophile and the carbonyl carbon a an electrophile B. The alpha carbon can gain a proton and act like an electrophile and the carbonyl carbon is a nucleophile C. The oxygen of the carbonyl group can attack the carbon of the carbonyl group D. Only esters can undergo self-condensation reactionsarrow_forward
- which has the greatest acidity? a. 2-hexanol b. valeric acid c. naphtol d. ethyl propionate which produces effervescence when dissolved in 10% sodium bicarbonate? a. 2-hexanol b. valeric acid c. napththol d. ethyl propionatearrow_forwardQUESTION 1 Which of the following compounds has an activating group? A. Benzoic acid B. None in the answer C. Bromobenzene D. Diphenylamine QUESTION 2 Which of the following compounds has a deactivating group attached to the benzene ring? A. Toluene B. Acetanilide C. Phenol D. Salicyclic Acidarrow_forward5. Consider the reaction below to answer the following questions: + H₂O a. vicinal diol b. geminal diol c. acetal d. ketal e. HO- hemiacetal HO a) Write the complete stepwise mechanism for the reaction shown above. Show all intermediate structures and all electron flow with arrows. OH b) The substance formed on addition of water to an aldehyde or ketone is called a hydrate or a/an:arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





