
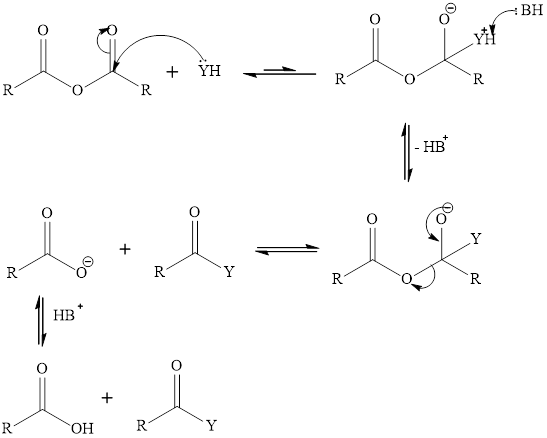
Interpretation:
To predict that out of alcohol, water, and
Concept introduction:
Acid anhydride can react with water, alcohol and amine. When acid anhydride reacts with water it gives two equivalent of
Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
Organic Chemistry (8th Edition)
- Describe a reaction mechanism that occurs to maleic acid when hydrochloric acid is added.arrow_forwardBy taking into account electronegativity differences, draw the products formed by heterolysis of the carbon–heteroatom bond in each molecule. Classify the organic reactive intermediate as a carbocation or a carbanion.arrow_forwardWrite the reactions and reactants benzoic acid chloride to benzoic anhydride benzoic anhydride to ethyl benzamide Write the reaction mechanism for the second reactionarrow_forward
- When propene reacts with gaseous hydrogen bromide, HBr, two products, 1-bromopropane and 2-bromopropane are formed. The reaction is a two-step process in which the electrophilic attack occurs in the first step. Identify the electrophile in this reaction Draw a diagram showing the first step of the reaction that leads to the production of 2-bromopropane.arrow_forwardSuggest a possible structure for Compound X.arrow_forwardExplain why pentane-2,4-dione forms two alkylation products (A and B) when the number ofequivalents of base is increased from one to two.arrow_forward
- Complete the following proposed acid–base reactions, and predict whether the reactants or products are favored.arrow_forwardAcetylcholine esterase is an important enzyme in neural synaptic signal transmission. It breaks down the neurotransmitter acetylcholine after it binds the acetylcholine receptor. Its active site has the same features used by serine proteases. Here is the reaction catalyzed by Acetylcholine esterase: Acetylcholine Choline OH Acetate + H Acetylcholine is an ester, while peptide bonds cleaved by proteases are amides. How should acetylcholine estease's mechanism accommodate this? This cannot be determined from the information presented in class and in this question. For amides, the amide nitrogen is deprotonate to make it more like an ester. The same mechanism cleaves deprotonated amides and esters. No changes are needed. The mechanism same works for esters and amides. Prior to cleavage, a transamination converts the ester to an amide. The esterase will make a covalent intermediate to an amine to create the amide to be cleaved.arrow_forwardChitosan is derived from the shells of crustaceans. it is only soluble in an aqueous acidic medium but not in organic solvents. When reacted with phthalic anhydride, it forms an amide. The product is no longer soluble in the aqueous medium but now is soluble in organic solvents. a) Draw the product of the reaction between chitosan and phthalic anhydride. b) Briefly explain the behaviour change in solubility of the starting material and the final product.arrow_forward
- Hydration of aldehydes and ketones can be catalyzed by acid or base. Bases catalyze hydration by: protonating the carbonyl oxygen making the carbonyl group more electrophilic employing hydroxide ion, which is a better nucleophile than water making the carbonyl group less electrophilic shifting the equilibrium position of the reaction to favor productsarrow_forwardWhich of the following will produce ammonia upon hydrolysis? "NH2arrow_forwardShow how acid derivatives hydrolyze to carboxylic acids under either acidic or basicconditions. Explain why some acid derivatives (amides, for example) require muchstronger conditions for hydrolysis than other derivatives.arrow_forward


 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning



