
(a)
Interpretation: The species which are used for the transesterification of esters other than acids has to be identified.
Concept introduction:
Transesterification is the process of formation of a new ester molecule from the reaction of alcohol and an ester. This is like hydrolysis of ester but here nucleophile is alcohol molecule instead of
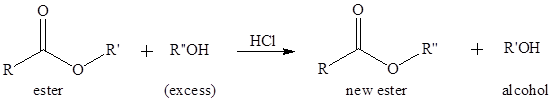
If a better nucleophile is used in place of alcohol molecule that will also increase the rate of transesterification reaction. The conjugate base of an alcohol is an alkoxide which is a better nucleophile and can attack the carbonyl carbon even its oxygen is not protonated.
(b)
Interpretation: The rate of aminolysis reaction of esters does not increase in presence of
Concept introduction: The reaction of a
Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
Organic Chemistry (8th Edition)
- Why is an alkylamine more basic than ammononia?arrow_forwardGive a reason why reduction reaction (with H2,pt) of nitrobenzene to alanine is so importantarrow_forwarda. A student did not obtain any ester when he added 2,4,6-trimethylbenzoic acid to an acidic solution of ethanol. Why? (Hint: Build models.)b. Would he have encountered the same problem if he had tried to synthesize the methyl ester of 4-methylbenzoic acid in the same way?arrow_forward
- The reaction of an ester with an amine is not as slow as the reaction of an ester with water or an alcohol. Explain with reason. Explain why the rate of aminolysis of an ester cannot be increased by H+, OH- or OR-. How can you activate the carboxylic acid? Is acid catalyzed hydrolysis of acetamide a reversible or an irreversible reaction. Explain.arrow_forwardDraw a good arrow-pushing mechanism for the basic hydrolysis of PLA to affordsodium lactate.arrow_forwardBy using only amide/Peptide bond forming rxn and a lKylation rxns. Provide all the reaction steps with the reagent needed and conditions with out showing the mechanism for each step to form the product. Note: Use protecting groups like (Boc, Fmoc, and others) H2NNH2+arrow_forward
- Arabitol is produced by the reduction of D‑arabinose. Modify the molecule to show the product of the reaction. Add or remove atoms or bonds as needed.arrow_forwardChemistry In a well-designed synthesis the Aldol reaction can produce only one product. However, if a chemist is not thoughtful while planning their experiments the Aldol reaction can produce MANY possible products. Explain two ways side products were avoided in the synthesis of dibenzylacetone. Hint: think steps in the procedure as well as the choice of starting materials.arrow_forwardWhy an amino group (NH2) directs electrophilic aromatic substitutionto the ortho and para positions ?arrow_forward
- When the pentaacetate anomer obtained in this reaction is heated with ZnCl2 and acetic anhydride, it is converted into the other anomer. Explain this observationarrow_forwardShow the Reaction of ROH with SOCl2 ?arrow_forwardHow to use streckers synthesis to prepare phenylalanine in the laboratoryarrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

