
Organic Chemistry (8th Edition)
8th Edition
ISBN: 9780134042282
Author: Paula Yurkanis Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 15, Problem 65P
Interpretation Introduction
Interpretation:
The role of
Concept introduction:
Transesterification is the process of formation of a new ester molecule from the reaction of alcohol and an ester. This is like hydrolysis of ester but here nucleophile is alcohol molecule instead of
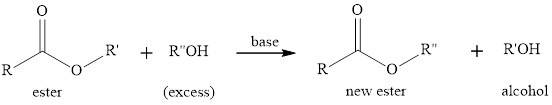
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Please correct answer and don't used hand raiting
this is an induced approximation. Can
u draw the mechanism ?
요
H
NaOH
1-1
OH
Please correct answer and don't used hand raiting
Chapter 15 Solutions
Organic Chemistry (8th Edition)
Ch. 15.1 - The aromas of many flowers and fruits are due to...Ch. 15.1 - Name the following:Ch. 15.1 - Prob. 3PCh. 15.2 - Which is longer, the carbon-oxygen single bond in...Ch. 15.2 - There are three carbon-oxygen bonds in methyl...Ch. 15.2 - Prob. 6PCh. 15.4 - a. What is the product of the reaction of acetyl...Ch. 15.4 - What is the product of an acyl substitution...Ch. 15.5 - a. Which compound has the stretching vibration for...Ch. 15.5 - Using the pKa values listed in Table 15.1, predict...
Ch. 15.5 - Is the following statement true or false? If the...Ch. 15.6 - Starting with acetyl chloride, what neutral...Ch. 15.6 - Prob. 13PCh. 15.7 - Starting with methyl acetate, what neutral...Ch. 15.7 - We saw that it is necessary to use excess amine in...Ch. 15.7 - Prob. 17PCh. 15.7 - Which ester hydrolyzes more rapidly? a. methyl...Ch. 15.7 - a. state three factors that cause the uncatalyzed...Ch. 15.8 - Prob. 21PCh. 15.8 - Using the mechanism for the acid-catalyzed...Ch. 15.8 - Prob. 23PCh. 15.8 - Show the mechanism for the acid-catalyzed...Ch. 15.8 - Prob. 25PCh. 15.8 - Write the mechanism for the acid-catalyzed...Ch. 15.8 - Write the mechanism for the acid-catalyzed...Ch. 15.9 - Prob. 28PCh. 15.9 - Prob. 29PCh. 15.10 - Show how each of the following esters could he...Ch. 15.10 - Prob. 32PCh. 15.11 - Prob. 33PCh. 15.11 - Which of the following reactions leads to the...Ch. 15.12 - Prob. 35PCh. 15.12 - Prob. 36PCh. 15.13 - Prob. 37PCh. 15.14 - Prob. 38PCh. 15.14 - Prob. 39PCh. 15.15 - Prob. 40PCh. 15.15 - Which alkyl halides from the carboxylic acids...Ch. 15.16 - Prob. 43PCh. 15.16 - Prob. 44PCh. 15.16 - Prob. 45PCh. 15.17 - Prob. 46PCh. 15.18 - How could you synthesize the following compounds...Ch. 15 - Prob. 48PCh. 15 - Name the following:Ch. 15 - Prob. 50PCh. 15 - What compound are obtained from the fallowing...Ch. 15 - a. Rank the following esters in order of...Ch. 15 - Because bromocyclohexane is a secondary alkyl...Ch. 15 - a. Which compound would you expect to have a...Ch. 15 - How could you use 1H NMR spectroscopy to...Ch. 15 - Rank the following compounds in order of...Ch. 15 - Prob. 57PCh. 15 - Prob. 58PCh. 15 - Prob. 59PCh. 15 - A compound with molecular formula C5H10O2 gives...Ch. 15 - Prob. 61PCh. 15 - Prob. 62PCh. 15 - Prob. 63PCh. 15 - Prob. 64PCh. 15 - Prob. 65PCh. 15 - Prob. 66PCh. 15 - Two products, A and B, are obtained from the...Ch. 15 - Prob. 68PCh. 15 - Prob. 69PCh. 15 - Prob. 70PCh. 15 - Prob. 71PCh. 15 - Prob. 72PCh. 15 - When treated with an equivalent of methanol,...Ch. 15 - a. Identify the two products obtained from the...Ch. 15 - Prob. 75PCh. 15 - Prob. 76PCh. 15 - a. When a carboxylic acid is dissolved in...Ch. 15 - Prob. 78PCh. 15 - Identity the major and minor products of the...Ch. 15 - When a compound with molecular formula C11H14O2...Ch. 15 - Prob. 81PCh. 15 - Prob. 82PCh. 15 - Prob. 83PCh. 15 - The 1H NMR spectra for two esters with molecular...Ch. 15 - Show how the following compounds could be prepared...Ch. 15 - Prob. 86PCh. 15 - Prob. 87PCh. 15 - The intermediate shown here is formed during the...Ch. 15 - Prob. 89PCh. 15 - Propose a mechanism that accounts for the...Ch. 15 - Catalytic antibodies catalyze a reaction by...Ch. 15 - Prob. 92P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Please correct answer and don't used hand raitingarrow_forwardPlease correct answer and don't used hand raitingarrow_forward(a) The following synthesis of the molecule shown in the circle has a major problem. What is this problem? (2 pts) 1) HBr (no peroxides) 2) H- NaNH2 Br 3) NaNH, 4) CH3Br 5) H2, Pd (b) Starting with the molecule shown below and any other materials with two carbons or less, write out an alternate synthesis of the circled molecule. More than one step is needed. Indicate the reagent(s) and the major product in all the steps in your synthesis. (5 pts) 2024 Fall Term (1) Organic Chemistry 1 (Lec) CHEM 22204 02[6386] (Hunter College) (c) Using the same starting material as in part (b) and any other materials win two carpons or less, write out syntheses of the circled molecules shown below. More than one step is needed in each case. Indicate the reagent(s) and the major product in all the steps in your synthesis. You may use reactions and products from your synthesis in part (b). (5 pts)arrow_forward
- alt ons for Free Response Questions FRQ 1: 0/5 To spectrophotometrically determine the mass percent of cobalt in an ore containing cobalt and some inert materials, solutions with known [Co?) are prepared and absorbance of each of the solutions is measured at the wavelength of optimum absorbance. The data are used to create a calibration plot, shown below. 0.90- 0.80- 0.70 0.60 0.50 0.40- 0.30 0.20- 0.10- 0.00- 0.005 0.010 Concentration (M) 0.015 A 0.630 g sample of the ore is completely dissolved in concentrated HNO3(aq). The mixture is diluted with water to a final volume of 50.00 ml. Assume that all the cobalt in the ore sample is converted to Co2+(aq). a. What is the [Co2] in the solution if the absorbance of a sample of the solution is 0.74? 13 ✗ b. Calculate the number of moles of Co2+(aq) in the 50.00 mL solution. 0.008 mols Coarrow_forwardPlease correct answer and don't used hand raitingarrow_forwardCloso-boranes and arachno-boranes are structures that exhibit B-B, B-H-B, and B-H bonds. Correct?arrow_forward
- Indicate why boron hydrides cannot form large linear or planar structures.arrow_forwardNido-boranes are structures with the molecular formula BnHn+4 that exhibit B-B, B-H-B and B-H bonds. Correct?arrow_forward8:07 AM Wed Dec 18 Final Exam 2024 copy Home Insert Draw Page Layout Formulas Data Review AA 田 General A G fx Alexis Cozort ☑ ⚫ 61% A B D E F H K M N P R S T U 3+ 10 125 mM that yielded peak heights of Aa = 9 1-(a)A sample solution was examined under XRF to quantify the analyte Ce³+. Find the response factor F, when standardized concentration of analyte [Ce³+]A = concentration of internal standard S i.e. [In³*]s = 151 mM was spiked with standardized 1600 and As = 3015 respectively? 11 12 (i)Define F, F = Aa As [A] [S] + X 13 (*Define with variables) 4000 14 15 (ii)Calculate F, F = numeral (You will use the F value in part 1-(b) below) As 16 (*Calculate with numerals) 17 18 1-(b)To determine the unknown conc of analyte [Ce³+], a volume of 15 mL of internal standard S having a concentration [In³+]s = 0.264 M 19 20 was added to 45 mL of unknown, and the mixture was diluted to 100 mL in a volumetric flask. XRF analysis yielded a spectrum, Figure-1, where peak heights A and As are…arrow_forward
- All structural types of Boron hydrides exhibit B-B, B-H-B and B-H bonds. Correct?arrow_forwardN-nitrosodimethylamine (NDMA) is a suspected carcinogen that can form via reactions between dimethylamine (DMA) and monochloramine (NH2Cl). The relevant elementary reactions and the corresponding rate constants are as shown below. Reaction Rate constant (M¹s¹) DMA + NH2Cl = DMCA + NH3 k =1.4×10-1, kr = 5.83×10-3 1.28×10-3 DMA + NH2Cl → UDMH UDMH + NH2Cl → NDMA -> 1.11×10-1 If the initial concentrations of DMA and NH2Cl are given, you should be able to predict the concentrations of all species at any given reaction time. Please write down the rate equations for DMA, NH2C1, DMCA, UDMH and NDMA.arrow_forwardYou wish to add enough NaOCl (sodium hypochlorite) to a 150 m³ swimming pool to provide a dose of 5.0 mg/L TOTOCI as Cl2. (a) How much NaOCI (kg) should you add? (Note: the equivalent weight of NaOCl is based on the reaction: NaOCl + 2H + 2 e→CI + Na +H₂O.) (10 pts) (atomic weight: Na 23, O 16, C1 35.5) (b) The pH in the pool after the NaOCl addition is 8.67. To improve disinfection, you want at least 90% of the TOTOCI to be in the form of HOCI (pKa 7.53). Assuming that HOCI/OCI is the only weak acid/base group in solution, what volume (L) of 10 N HCl must be added to achieve the goal? (15 pts) Note that part a) is a bonus question for undergraduate students. If you decide not to work on this part of the question, you many assume TOTOCI = 7×10-5 M for part b).arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning


Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning
Characteristic Reactions of Benzene and Phenols; Author: Linda Hanson;https://www.youtube.com/watch?v=tjEqEjDd87E;License: Standard YouTube License, CC-BY
An Overview of Aldehydes and Ketones: Crash Course Organic Chemistry #27; Author: Crash Course;https://www.youtube.com/watch?v=-fBPX-4kFlw;License: Standard Youtube License