
Concept explainers
Interpretation: Starting material and reagents required to prepare the given epoxide need to be identified.
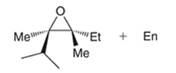
Concept introduction:
Preparation of epoxide-
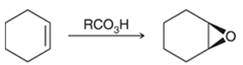
Peroxy acids generally used in this process are MCPBA and peroxyacetic acid. The formation of epoxide via peroxy acid is a stereospecific process; thus, cis substituents in alkene (starting material) remain at cis to each other in the epoxide (product). Similarly, trans substituents in alkene remain at trans to each other.
The preparation of epoxides from halohydrins can simply take place upon treatment with a strong base. Here, halohydrin can be formed from alkene after treatment with a halogen in the presence of water.
The reactions involved in the preparation are as follows
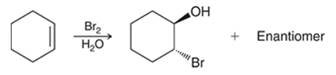
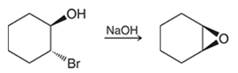
The above reaction takes place via intramolecular
Want to see the full answer?
Check out a sample textbook solution
Chapter 13 Solutions
EBK ORGANIC CHEMISTRY-PRINT COMPANION (
- Claisen rearrangement of an allyl phenyl ether with substituent groups in both ortho positions leads to the formation of a para-substituted product. Propose a mechanism for the following rearrangement.arrow_forwardClaisen condensation between diethyl phthalate and ethyl acetate followed by saponification, acidification, and decarboxylation forms a diketone, C9H6O2. Propose structural formulas for compounds A and B and the diketone.arrow_forwardTreatment of 1-aminoadamantane, C10H17N, with methyl 2,4-dibromobutanoate in the presence of a nonnucleophilic base, R3N, involves two successive SN2 reactions and gives compound A. Propose a structural formula for compound A.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
 EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT


