
Concept explainers
(a)
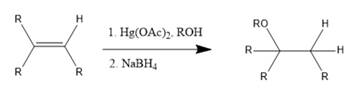
Interpretation: The reagents required to prepare given ether via an alkoxymercuration-demercuration need to be identified.

Concept Introduction: In alkoxymercuration-demercuration reaction, ethers are prepared from
The general reaction is represented as follows:
(b)
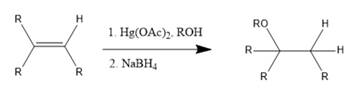
Interpretation: The reagents required to prepare given ether via an alkoxymercuration-demercuration need to be identified.
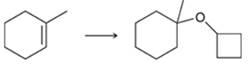
Concept Introduction: In alkoxymercuration-demercuration reaction, ethers are prepared from alkene in which Markovnikov addition of alcohol takes place across an alkene. Here, -OR group is placed at a more substituted position and H is placed at a less substituted position.
The general reaction is represented as follows:
Want to see the full answer?
Check out a sample textbook solution
Chapter 13 Solutions
EBK ORGANIC CHEMISTRY-PRINT COMPANION (
- Draw reaction mechanisms with all reactants, arrows, intermediates, and products. Your mechanism must account for all the products if more than one product is formed. 4-methycyclohexanol with phosphoric acid H3PO4 to for 1-methycyclohexene, 3- methylcyclohexene and 4-methycyclohexenearrow_forwardWhat are the major products obtained when each of the following ethers is heated with one equivalent of HI?arrow_forwardUsing your reaction roadmap as a guide, show how to convert cyclohexane into hexanedial. Show all reagents and all molecules synthesized along the way.arrow_forward
- 1. How would you prepare the following ethers?arrow_forwardGive two sets of reactants (each set including an alkyl halide and a nucleophile) that could be used to synthesize the following ether:arrow_forwardFrom the list of available starting materials, select the reagents you would use to best prepare the following compound by Williamson ether synthesisarrow_forward
- Provide a synthetic route for the following transformation: Please provide steps and type of reactions as well as using commercially available reagents, with an explanation of steps. OH N. Harrow_forwardSelect two reagents that will synthesize the following ether using Williamson ether synthesis:arrow_forwardB) Write the most stable enol forms of the following compound, and choose which of them is more stal With explanation. TH1003 al sein) soobel. Xudda0 Jor'larizzA suddz zuddall Jen CH3arrow_forward
- Choose the most correct set of reagents for the following reaction.arrow_forwardb) What starting materials are needed to prepare the compound below by intramolecular Michael reaction? c) Show how you would camy out the following transformations. OHarrow_forwardProvide all the reagents and show all structures of the product formed in each step, not just a list of reagents. b) Benzene c) Toluene OHC Ĵ Ĉ COOHarrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
