
(a)
Interpretation:
The product should be identified for the given reaction.
Concept introduction:
Acetals: Generally Acetals are used to protect the
In this reaction acetone is protected as acetal by using ethylene glycol. Generally acetals are less stable compound.
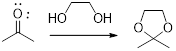
When a ketone reacts with a
(b)
Interpretation:
The product should be identified for the given reaction.
Concept introduction:
Acetals: Generally Acetals are used to protect the ketone and aldehyde (carbonyl group).
In this reaction acetone is protected as acetal by using ethylene glycol. Generally acetals are less stable compound.
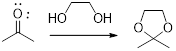
When a ketone reacts with a diol, the formation of cyclic ketal takes place. The reaction takes place in the presence of an acid catalyst. An acid catalyst is to activate the carbonyl group. The nucleophile attacks the carbonyl group leading to the formation of oxonium ion. The oxonium ion is again attacked by the nucleophile to form the cyclic ketal. The cyclic ketal can be five-memebered or six-memebered depending upon the diol used in the reaction.
Want to see the full answer?
Check out a sample textbook solution
Chapter 12 Solutions
Essential Organic Chemistry, Global Edition
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





