
(a)
Interpretation:
The species which are used for the transesterification of esters other than acids has to be identified.
Concept introduction:
Transesterification is the process of formation of a new ester molecule from the reaction of alcohol and an ester. This is like hydrolysis of ester but here nucleophile is alcohol molecule instead of
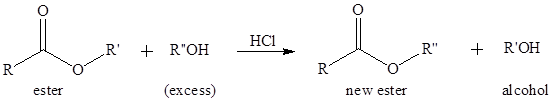
If a better nucleophile is used in place of alcohol molecule that will also increase the rate of transesterification reaction. The conjugate base of an alcohol is an alkoxide which is a better nucleophile and can attack the carbonyl carbon even its oxygen is not protonated.
(b)
Interpretation:
The rate of aminolysis reaction of esters does not increase in presence of
Concept introduction:
The reaction of a
Trending nowThis is a popular solution!

Chapter 11 Solutions
Essential Organic Chemistry, Global Edition
- 2. Draw in the product (or products) formed from the acidic hydrolysis of the following compound. ^^ Hydrolysisarrow_forwardWhich amino acids on proteins can form glycosidic linkages? Select one: a. Cysteine and methionine b. Tryptophan and phenylalanine c. Glycine and proline d. Serine and asparagine e. Alanine and leucinearrow_forwardWhat is the difference between the aminolysis of an ester and that of an acyl halide?arrow_forward
- If an acetyl group is attached to the sulfhydryl group of coenzyme A (shown below), what new functional group is formed? NH2 N. Cofofe OHC CH HS N. O OH phosphoanhydride linkage ester linkage O amido group O carboxyl group O carbonyl group O thioester linkage O ether linkage O phosphoester linkage O amino grouparrow_forwardB. FATS AND OILS Consider the structure of triglyceride, SABON. -(CH₂)14CH3 -(CH₂) 16CH3 (CH₂)14CH3 SABON 1. Is SABON a simple or mixed triglyceride? 2. Draw the structures of the products produced from the acid hydrolysis of SABON in your answer sheet.arrow_forwardIdentify the ester product of the reaction. HC HC IU OT CH OH + MC- OH H+, heat HC HC. HC جدید IL HC. CH HC علام HC. CH CH | + H2O CH₂ CH₂ CH3arrow_forward
- Indicate the INCORRECT alternative: a) The hydrolysis of the esters is carried out with heating to boiling and in an acidic or basic medium. b) In the formation of cyanohydrins, a molecule of HCN is added to an aldehyde or ketone. c) The formation of imines is carried out much more quickly in an extremely acidic medium (pH=1.0) d) Esters are more reactive than amides. e) Acetals are the product of the addition of two alcohol molecules to an aldehyde or ketone.arrow_forwardDraw the product of ethyl acetoacetate, base, (R)-2-bromobutane, hydrolysis and heat. Draw the product of diethyl hexanedioate, base, hydrolysis and heat.arrow_forwardfid the molecular structure of 1. deprotonated form of 2-amino-3hydroxypropanoic acid 2.zwitterionic form of 2-Amino-3-methylpentanoic acid 3. protonated form of 2-Amino-3-methylpentanoic acid 4. deprotonated form of 2,5 diamino5-oxopentanoic acid 5. zwitterionic form of 2-amino-3hydroxypropanoic acid 6.2-phenyl propanol 7. 2,4,6-trinitrophenol 8. oxoheptanoic acid 9. (4E)-2,6-dimethylhepta2,4-dien3ol 10. 3-methylbutan1-olarrow_forward
- Explain why amino acids, unlike most amines and carboxylic acids, are insoluble in diethyl ether.arrow_forwarda) Esters can be cleaved back into a carboxylic acid and an alcohol by reaction with water and a base. The reaction is called a saponification from the Latin sapo which means soap. The name comes from the fact that soap used to be made by the ester hydrolysis of fats. Due to the basic conditions a carboxylate ion is made rather than a carboxylic acid. i) Write the reactions of saponification ii) Write the reaction of saponification mechanism of the reaction.arrow_forwardThe carbonyl group on D-glucose can be reduced to d-glucitol. Identify the reagents that can chrry out this reduction. A.water in acidic solution B. hydrogen with a Platinum catalyst (Hy/Pt) C. Sodium dichromate (NaCr2O) in acidic solution D. Benedict’s reagent E. Enzymesarrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
