
(a)
Interpretation:
Considering the acid catalyzed mechanism of ester hydrolysis reaction answer the following,
The species which can be represented by
Concept Introduction:
An acid catalyzed hydrolysis of ester is much faster reaction as compared to uncatalyzed hydrolysis of ester. The acid catalyzed reaction mechanism is written as,
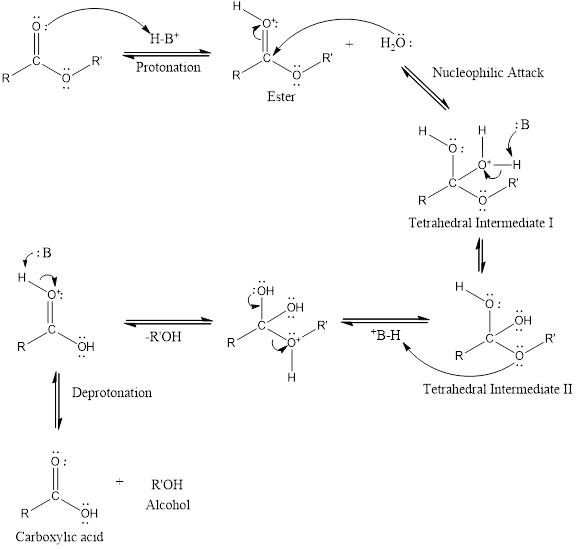
(b)
Interpretation:
Considering the acid catalyzed mechanism of ester hydrolysis reaction answer the following,
The species which can be represented by
Concept Introduction:
An acid catalyzed hydrolysis of ester is much faster reaction as compared to uncatalyzed hydrolysis of ester. The acid catalyzed reaction mechanism is written as,
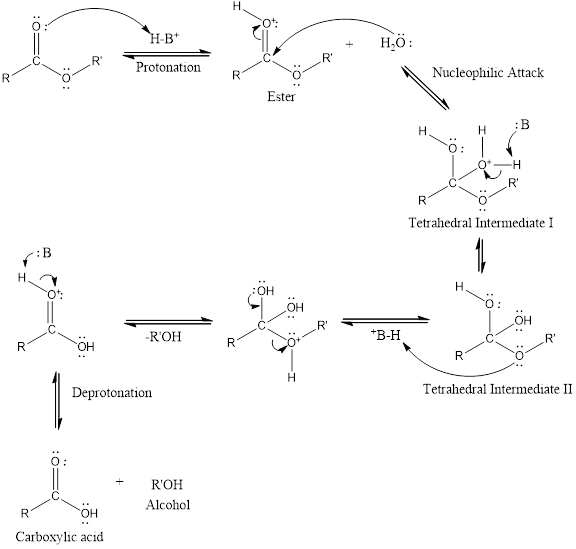
(c)
Interpretation:
Considering the acid catalyzed mechanism of ester hydrolysis reaction answer the following,
The species which is mostly used as
Concept introduction:
An acid catalyzed hydrolysis of ester is much faster reaction as compared to uncatalyzed hydrolysis of ester. The acid catalyzed reaction mechanism is written as,

(d)
Interpretation:
Considering the acid catalyzed mechanism of ester hydrolysis reaction answer the following,
The species which is mostly used as
Concept Introduction:
An acid catalyzed hydrolysis of ester is much faster reaction as compared to uncatalyzed hydrolysis of ester. The acid catalyzed reaction mechanism is written as,

Want to see the full answer?
Check out a sample textbook solution
Chapter 11 Solutions
Essential Organic Chemistry, Global Edition
- 5. a. Clearly identify the most acidic H atom in each of the structures shown below. Circle or draw it on the structure. ůl A B C b. Which of the compounds in part a can be completely deprotonated by NaOH? c. Which of the compounds in part a can be completely deprotonated by LDA? OHarrow_forwardWhich of the following anions is the most nucleophilic in polar protic solvents? a. F- b. Cl- C. Br- d. I- O a. fluoride b. chloride O c. bromide O d. iodidearrow_forwardWhich of the following is most likely to act as a base rather than a nucleophile? I II II IV а. I b. II с. Ш d. IVarrow_forward
- 31 What is the rate-determining step in the alkaline hydrolysis of an amide (reaction shown below) NH₂ H₂O/OH- he at + A. Bonding of OH- nucleophile to carbonyl carbon. B. Ejection of NH₂ from a tetrahedral intermediate. C. Protonation of an -NH₂ group in a tetrahedral intermediate. D. Ejection of NH3 from a tetrahedral intermediate. NH3arrow_forwardIn the following reaction which species acts as the nucleophile? OCH CH3 (H.C).NOCH CH CH,CH OH OCH,CH , OCH,CH, A. OCH,CH, (H,C),N B. C. CH,CH,OH D. (D)arrow_forwardWhich one of the following synthetic routes gives the best yield of meta-bromoaniline starting with benzene? NH2 HNO, 1) Sn. HCI Br, A. FeBr, H,SO, 2) NaOH Br metabromoaniline 1) Sn, HCI Br, HNO, В. H,SO, 2) NaOH FeBr, HNO, Br, 1) Sn, HCI C. FeBr H,SO, 2) NaOH 1) Sn, HCI HNO Br, D. 2) NaOH HSO FeBr, O A O B O C Ouestion 14 6 pts MacBook Pro Search or type URLarrow_forward
- ol de C 3. Caffeine is an alkaloid, meaning alkali- or base-like. Circle functional groups in the molecule which would make react as a base. CH3 CH3 a. O b. O 4. Write the equations that account for what happens in the hydrolysis of acetamide O || (CH₂C-NH₂) in (a) acid and in (b) base. woled mode) N I T CH3 H Caffeine custom page 114arrow_forwardWhat type(s) of catalysis is/are present in the enzymatic reaction below? SCOA H-His CH SCOA Asp CH COASH CH: CO HO-C-COO" HO-C-COO H-CH, acetyl-CeA SCOA CH, CH; CH COO COO COO oxaloacetate an intermediate citrate Select one or more: a. general acid b. general base C. metal ion d. specific base e. nucleophilic f. specific acidarrow_forwardIdentify the products obtained by the hydrolysis of the given compound. оо | | -COC- Select one: O a. one molecule of dibenzoic acid and one molecule of benzene O b. one molecule of benzene and one molecule of a carboxylic acid O c. two molecules of phenol O d. two molecules of benzoic acidarrow_forward
- Which option is the major product expected for the reaction below? a. b. C. d. CHO NaOH (trace) H₂O o HO OH OH OH CHO or ?arrow_forwardIn what way(s) does the carbon-group-bonded-to metal of an organometallic compound behave? Select one: ⒸA Oxidizing agent O O B. Acid (proton donor) C. Base (proton acceptor) D. Carbon nucleophile E. Carbon electrophile Both C and D Which of the following is NOT an organometallic compound?arrow_forwardIn the acid catalyzed halogenation of ketones, what is the rate determining step? A. The first step: protonation of the ketone. B. The very last step in the mechanism, namely, loss of a hydrogen ion to form the product. C. Loss of a proton from the alpha-carbon of the protonated ketone to form an enol intermediate. D. Attachment of a halogen molecule to the enol intermediate.arrow_forward
 EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

