
(a)
Interpretation:
The expected products obtained from the given reaction have to be predicted.
Concept introduction:
Esterification reaction and intermolecular esterification reactions:
General scheme for esterification reaction:
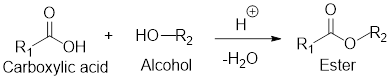
If the carboxylic acid and alcohol
Example for intermolecular esterification reaction:
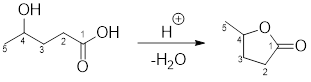
(b)
Interpretation:
The expected products obtained from the given reaction have to be predicted.
Concept introduction:
Transesterification and intermolecular transesterification reactions:
In the presence of an acid, ester and alcohol forms another new ester which is known as transester and a new alcohol will also be formed. This type of reaction is called transesterification reaction.
General scheme for transesterification reaction:
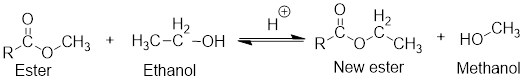
If the ester and alcohol are present within the same compound, then cyclic ester is formed which is known as lactone and the reaction is known as intermolecular transesterification reaction.
Example for intermolecular transesterification reaction:
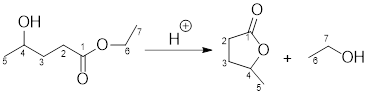
Want to see the full answer?
Check out a sample textbook solution
Chapter 11 Solutions
Essential Organic Chemistry, Global Edition
- Provide a step-by-step mechanism to account for the product of each of the following reactions. Show the structure of each of the intermediates and use curved arrows to indicate electron flow in each of these steps.arrow_forwardWhat is the major product obtained from the reaction of HBr with each of the following?arrow_forwardWrite the expected substitution product(s) for each reaction and predict the mechanism by which each product is formedarrow_forward
- Which of the reaction conditions could afford the following transformation?arrow_forwardProvide a step-by-step mechanism to account for the product of the following reaction. Show the structure of each of the intermediates and use curved arrows to indicate electron flow in each of these stepsarrow_forwardpredict the product for each of the following reactions and propose a mechanism that explains the formation of each product ?arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





