
Concept explainers
(a)
Interpretation: The alkyl halide that is used to form the given
Concept introduction: The reaction of
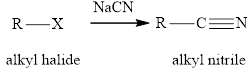
The hydrolysis reaction of alkyl nitrile in presence of acid is written as:
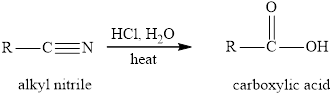
(b)
Interpretation: The alkyl halide that is used to form the given carboxylic acid has to be identified.
Concept introduction: The reaction of alkyl halides with sodium cyanide gives alkyl cyanides or nitriles. After the reaction there is increase incarbon atoms of the alkyl chain of alkyl halide. The nitriles obtained undergo hydrolysis reaction in presence of acid and gives carboxylic acid. The general reaction of alkyl halides and sodum cyanide is written as:
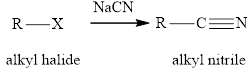
The hydrolysis reaction of alkyl nitrile in presence of acid is written as:
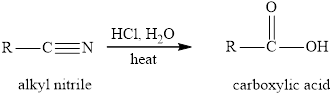
(c)
Interpretation: The alkyl halide that is used to form the given carboxylic acid has to be identified.
Concept introduction: The reaction of alkyl halides with sodium cyanide gives alkyl cyanides or nitriles. After the reaction there is increase incarbon atoms of the alkyl chain of alkyl halide. The nitriles obtained undergo hydrolysis reaction in presence of acid and gives carboxylic acid. The general reaction of alkyl halides and sodum cyanide is written as:
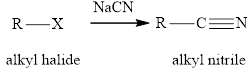
The hydrolysis reaction of alkyl nitrile in presence of acid is written as:
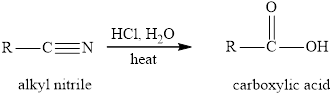
Want to see the full answer?
Check out a sample textbook solution
Chapter 11 Solutions
Essential Organic Chemistry, Global Edition
- List the following compounds in order of increasing water solubility: a.ethoxyethane b.propanoic acid c.pentane d.1 butanolarrow_forwardWhat reagents/conditions would you use to convert a Alkyl Benzene to a carboxylic acid?arrow_forwardWhat reaction occurs when a strong base is added to a carboxylic acid?arrow_forward
- 17. Which functional groups are in the following molecule of aspirin? HO, a benzene ring, an ester, a ketone, and an alcohol b. a benzene ring, two ketones, an ether, and an alcohol a benzene ring, a carboxylic acid, an ether, and a ketone d. a benzene ring, a carboxylic acid, and an ester a. c.arrow_forward1. What are the common derivatives of carboxylic acids? How are they named? 2. What reactions do carboxylic acid derivatives undergo?arrow_forwardWhich of the following carboxylic acid is toxic when ingested? a.Citric acid b.Malic acid c.Acetic acid d.Oxalic acid What is the general formula for Grignard reagents? a.RCOOH b.RMgX c.RX d.RCOX Oxidation of alkyl benzenes in the presence of Sulfuric acid will produce which carboxylic acid? a.Cyclohexanecarboxylic acid b.Benzoic acid c.Acrylic acid d.Fumaric acid What dicarboxylic acid contains six carbon atoms in its structure? a.Capric acid b.Pimelic acid c.Adipic acid d.Hexanoic acid Which of the following is an example of an unsaturated carboxylic acid? a.Succinic acid b.Formic acid c.Glycolic acid d.Acrylic acidarrow_forward
- Carboxylic acids are weak acids and dissociate in water. What species is formed by the dissociation of benzoic acid in aqueous solution? A) benzene B) benzoate ion C) benzaldehyde D) phenol E) toluenearrow_forward45. Which of the following compounds has the lowest water solubility? a. butanal b. heptanal c. hexanal d. pentanal 48. Which of the following compounds is the most soluble in water? a. butanal b. heptanal c. hexanal d. pentanal 56. Which of the following compounds will undergo oxidation using potassium dichromate to form a carboxylic acid? A a. A and B only b. A and C only c. B only d. C only H B o Carrow_forwardWhy are carbonyl compounds considered weakly acidic? Would you expect carbonyl compounds to be more acidic than alkanes? Explain.arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER




