
Concept explainers
Interpretation:
The difference between ether and ester
Concept introduction:
Organic molecules have some structural features in addition to
Table given below gives information about some common functional groups:
| Type of Compound | General Structure |
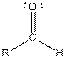 | |
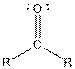 | |
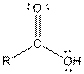 | |
| Ester | 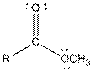 |
| Amide | 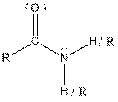 |
| Alcohol |  |
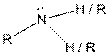 | |
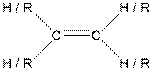 | |
| Ether | 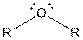 |
Here, R = any carbon backbone
Ball and stick model of molecule is determined by X-ray crystallography, in which the hydrogen atoms are depicted as white balls, carbon atoms as black balls, nitrogen atoms as blue balls and oxygen atom as red balls.
Want to see the full answer?
Check out a sample textbook solution
Chapter 11 Solutions
General, Organic, and Biological Chemistry - 4th edition
- The general formula of an alkane is CnH2n+2 . What is the general formula of an (a) alkene? (b) alkyne? (c) alcohol derived from an alkane?arrow_forwardDraw general formulas for an alcohol and phenol, showing the functional group.arrow_forwardSelect all functional groups present in the following structure of a drug commonly found in a dentist's office N a) Alkyl halide b) Alcohol c) Carboxylic acid d) Phenol e) Amine f) Ether Og) Amide h) Ester i) Ketone 0 j) Aromatic ring (aka phenyl) (aka arene)arrow_forward
- Differentiate Cyclohexane vs Benzenearrow_forwardIdentify the type of compounds (aldehyde, ketone, alcohol, phenol, ester, etc) shown in the imagearrow_forwardHO НО,, H O alcohol, ester, amine, ether alcohol, ester, amine, ether Batrachotoxin is a poison found in some species of frogs. What functional group/s is/are present in Batrachotoxin? Alcohol, carboxylic acid,amine, ether || alcohol, ketone, amine, ether N O NHarrow_forward
- How can phenol be distinguished from cyclohexanol? O A. solubility in water B. solubility in hydrochloric acid solution C. solubility in sodium bicarbonate solution D. solubility in sodium hydroxide solutionarrow_forwardGive the different properties between Aldehydes and Ketones.arrow_forwardWhat kind of alcohol is this?arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning





