
General, Organic, and Biological Chemistry - 4th edition
4th Edition
ISBN: 9781259883989
Author: by Janice Smith
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 11.4, Problem 11.7PP
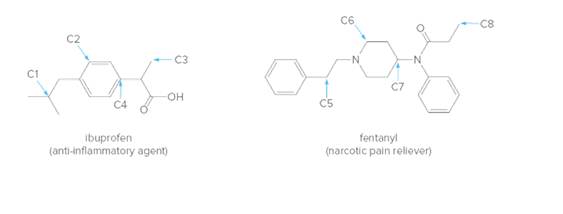
How many H’s are bonded to each indicated carbon (C1-C8) in the following drugs?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
How many non-equivalent carbons exist in the following compound? (Give a number)
CH3
H,C
Ethambutol is an anti-tuberculosis medicinal agent with two stereogenic centers, as shown in the given skeletal structure. It has 15, 2S configuration at its
stereogenic centers.
Redraw ethambutol, showing the correct configuration at each of the stereogenic centers.
OH
H
fil
N
H
2
OH
-
Click and drag to start drawing
a structure.
C C
С
X
Ś
CX
è
Which of the following exhibit C-H hypeconjugation only?
NH2
А
В
D
A and D
A and C
A
D
Chapter 11 Solutions
General, Organic, and Biological Chemistry - 4th edition
Ch. 11.1 - Prob. 11.1PCh. 11.2 - Fill in all H's and lone pairs in each compound.Ch. 11.3 - Prob. 11.2PPCh. 11.3 - Prob. 11.2PCh. 11.3 - Prob. 11.3PCh. 11.3 - Prob. 11.3PPCh. 11.3 - How many lone pairs are present in lidocaine, the...Ch. 11.4 - Convert each compound to a condensed formula.Ch. 11.4 - Convert each condensed formula to a complete...Ch. 11.4 - Convert each skeletal structure to a complete...
Ch. 11.4 - Prob. 11.5PCh. 11.4 - How many H’s are bonded to each indicated carbon...Ch. 11.4 - Using the skeletal structure, determine the...Ch. 11.5 - Prob. 11.7PCh. 11.5 - Prob. 11.8PCh. 11.5 - For each compound. [1] Identify the functional...Ch. 11.5 - How do a carboxylic acid and an alcohol differ?...Ch. 11.5 - Label each of the following condensed structures...Ch. 11.5 - Prob. 11.11PCh. 11.5 - Prob. 11.12PCh. 11.5 - Identify all of the functional groups in atenolol,...Ch. 11.5 - Prob. 11.13PCh. 11.5 - Prob. 11.10PPCh. 11.5 - Prob. 11.14PCh. 11.6 - Indicate the polar bonds in each compound. Label...Ch. 11.6 - Prob. 11.11PPCh. 11.6 - Prob. 11.16PCh. 11.6 - Predict the water solubility of each compound.Ch. 11.6 - Prob. 11.17PCh. 11.7 - Prob. 11.18PCh. 11.7 - Prob. 11.19PCh. 11.7 - Prob. 11.20PCh. 11 - Prob. 21PCh. 11 - Prob. 22PCh. 11 - Complete each structure by filling in all H’s and...Ch. 11 - Complete the structure of mepivacaine by filling...Ch. 11 - Prob. 25PCh. 11 - Prob. 26PCh. 11 - Prob. 27PCh. 11 - Prob. 28PCh. 11 - “Ecstasy” is a widely used illegal stimulant....Ch. 11 - Prob. 30PCh. 11 - Explain why each C—C—C bond angle in benzene...Ch. 11 - Prob. 32PCh. 11 - Convert each compound to a condensed structure.Ch. 11 - Convert each compound to a condensed structure.Ch. 11 - Convert each compound to a skeletal structure.Ch. 11 - Convert each compound to a skeletal structure.Ch. 11 - Convert each shorthand structure to a complete...Ch. 11 - Convert each shorthand structure to a complete...Ch. 11 - Convert each skeletal structure to a complete...Ch. 11 - Convert each skeletal structure to a complete...Ch. 11 - A and B are ball-and-stick models of two compounds...Ch. 11 - Prob. 42PCh. 11 - What is wrong in each of the following shorthand...Ch. 11 - Prob. 44PCh. 11 - Prob. 45PCh. 11 - Albuterol (trade names Proventil and Ventolin) is...Ch. 11 - Prob. 47PCh. 11 - Prob. 48PCh. 11 - Prob. 49PCh. 11 - (a) Identify the functional groups in donepezil,...Ch. 11 - Prob. 51PCh. 11 - GHB is an addictive, illegal recreational drug...Ch. 11 - Prob. 53PCh. 11 - Prob. 54PCh. 11 - Prob. 55PCh. 11 - Prob. 56PCh. 11 - Prob. 57PCh. 11 - (a) Identify the functional groups in venlafaxine,...Ch. 11 - You are given two unlabeled bottles of solids, one...Ch. 11 - State how potassium iodide (KI) and pentane...Ch. 11 - The given beaker contains 100 mL of the organic...Ch. 11 - Prob. 62PCh. 11 - Why do we need to know the shape of a molecule...Ch. 11 - 1,1-Dichloroethylene (CH2=CCl2) is a starting...Ch. 11 - Indicate the polar bonds in each molecule. Label...Ch. 11 - Indicate the polar bonds in each molecule. Label...Ch. 11 - Classify each molecule as polar or nonpolar.Ch. 11 - Classify each molecule as polar or nonpolar. a....Ch. 11 - Which molecule is more water soluble? Explain.Ch. 11 - Explain why pantothenic acid, vitamin B5, is water...Ch. 11 - Prob. 71PCh. 11 - Prob. 72PCh. 11 - Explain why regularly taking a large excess of a...Ch. 11 - You can obtain the minimum daily requirement of...Ch. 11 - Prob. 75PCh. 11 - Vitamin B6 is obtained by eating a diet that...Ch. 11 - Prob. 77PCh. 11 - Can an oxygen-containing organic compound, have...Ch. 11 - Prob. 79PCh. 11 - Prob. 80PCh. 11 - Benzocaine is the active ingredient in topical...Ch. 11 - Methyl salicylate is responsible for the...Ch. 11 - Answer the following questions about aldosterone,...Ch. 11 - Answer the following questions about...Ch. 11 - Prob. 85PCh. 11 - Skin moisturizers come in two types, (a) One type...Ch. 11 - THC is the active component in marijuana (Section...Ch. 11 - Cocaine is a widely abused, addicting drug....
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Provide a systematic name (IN SMALL LETTERS except for E/Z and R/S, NO SPACES) for each of the following compounds.arrow_forwardHow many degrees of unsaturation are present in following compound? C6H6arrow_forwardWhich of the following pairs are constitutional isomers? ClCH2CH2CH2CH3 and CH3CH2CH2CH2Cl H2NCH2CH=CHCOOH and HOOCCH=CH(H2N)CH2 CH3CH(OH)CH2CH3 and CH3CH2(HO)CHCH3 CH3CH=CHCH2OH and CH3CH=CHOCH3 CH3OCH2CH2CH2CH3 and CH3OCH2CH2CH2OCH3arrow_forward
- Label each C-C double bond in Kavain, a naturally occurring relaxant, as E or Z. II Kavain O I = Z and || = Z O I = E and I| = E OI = Z and I| = E O1=E and II = Zarrow_forwardHow many asymmetric carbons are present in the compound below? CH₂ $ (CH₂)₂CH 02 3 WHarrow_forwardPage of 11 ZOOM + A1. What is the arrangement of the bonds surrounding carbon in a saturated hydrocarbon? linear trigonal planar tetrahedral В trigonal bipyramidal A2. What is the definition of a constitutional isomer? A molecules with the same numbers and kinds of atoms arranged in different ways molecules with the same arrangements of atoms, but different arrangements of electrons molecules with the same connectivities, but different 3-D angles molecules with different isotopes of the same atoms В АЗ. How many hydrogen atoms does the following molecule contain? 12 В 16 18 Which functional group gives rise to a broad IR absorption between 3200 cm-1 and 3600 cm-1? A4. A O-H stretch C-H stretch O-H bend C-O stretch А5. How many resonances will appear in the 13C NMR spectrum of cyclopentanone? 3 4 Section A continues on the next page N468 ABCD ABCD ABCDarrow_forward
- Rank the following compounds according to increasing positive character of the carbon atom. least positive C Most positive C CH3F CH3OH CH3Li CH3I CH3CH3 CH3NH2arrow_forwardAre the following two structures the same compound, meaning are they conformers? CH₂CH3 HCH₂CH3 CH3 H H and H7 CH3 -H 'Harrow_forwardWhat is true about the relationship between the following compounds? CH3 CH3 CH; CH; CH3 CH3 A В O All three are stereoisomers of each other. OA and B are the same compound while C is a stereoisomer. All three are the same compound. A and B are the same while C is a conformer, all of the same compound . O A and C are the same while B is a stereoisomer.arrow_forward
- 8. Two structures of Lipitor (a drug used to lower cholesterol) are shown below. (a) Determine the absolute configuration of each indicated stereocenter. Fill in the correct circle. (b) Determine if the two structures are the same compound or stereoisomers. Fill in the correct circle. (a) НО. Carbon a HO O OH Carbon a: OR OS Carbon b H N Carbon b: R OS of H Carbon c: OR OS OH OH Carbon c F Carbon d: R OS OH Carbon d (b) The two structures are: O the same compound O stereoisomersarrow_forwardLabel each asymmetrical carbon in the compound below as Ror S. H3C Н НО ОНС НО Н CH OHarrow_forwardн н, н Identify which C-H bonds are axial and which are equatorial in the following Newman projection for cyclohexane. H H2 H エエarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
Lipids - Fatty Acids, Triglycerides, Phospholipids, Terpenes, Waxes, Eicosanoids; Author: The Organic Chemistry Tutor;https://www.youtube.com/watch?v=7dmoH5dAvpY;License: Standard YouTube License, CC-BY