
Concept explainers
(a)
Interpretation:
Whether the following molecule is polar or non-polar should be determined:
Concept introduction:
Polarity of any individual bond is determined by the electro-negativities of the atoms forming a bond. Any covalent bond is non-polar when two identical atoms or of similar electro-negativity are bonded together.
(b)
Interpretation:
Whether the following molecule is polar or non-polar should be determined:
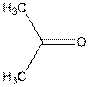
Concept introduction:
Polarity of any individual bond is determined by the electro-negativities of the atoms forming a bond. Any covalent bond is non-polar when two identical atoms or of similar electro-negativity are bonded together.
(c)
Interpretation:
Whether
Concept introduction:
Polarity of any individual bond is determined by the electro-negativities of the atoms forming a bond. Any covalent bond is non-polar when two identical atoms or of similar electro-negativity are bonded together.
(d)
Interpretation:
Whether
Concept introduction:
Polarity of any individual bond is determined by the electro-negativities of the atoms forming a bond. Any covalent bond is non-polar when two identical atoms or of similar electro-negativity are bonded together.
Want to see the full answer?
Check out a sample textbook solution
Chapter 11 Solutions
General, Organic, and Biological Chemistry - 4th edition
- Successive substitution of F atoms for H atoms in the molecule NH3 produces the molecules NH2F, NHF2, and NF3. a. Draw Lewis structures for each of the four molecules. b. Using VSEPR theory, predict the geometry of each of the four molecules. c. Specify the polarity (polar or nonpolar) for each of the four molecules.arrow_forwardUse the Molecule Shape simulator (http://openstaxcollege.org/I/6MolecShape) to build a molecule. Starting with the central atom, click on the double bond to add one double bond. Then add one single bond and one lone pair. Rotate the molecule to observe the complete geometry. Name the electron group geometry and molecular structure and predict the bond angle. Then click the check boxes at the bottom and right of the simulator to check your answers.arrow_forwardIs the Cl2BBCl2 molecule polar or nonpolar?arrow_forward
- Using the symbols and +, indicate the direction of polarity in each polar covalent bond. (a) CN (b) NO (c) CClarrow_forwardArrange the single covalent bonds within each set in order of increasing polarity. (a) CH, O H, NH (b) CH, BH, OH (c) CH, CCl, CI (d) CS, CO, CN (e) CLi, CB, CMgarrow_forwardChoose the bond that is the least polar. Explain your reasoning P—S d. Sr—O C—O e. Fe—P N—Narrow_forward
- 5. Complete the chart for the molecular compounds listed. Molecule Lewis Name of Bond angle Polarity (Polar/Non polar) structure shape SiBr4 XeF3arrow_forward1- What type of chemical bond is expected between the given pair of atoms? Justify your answers a. Potassium and chlorine b. bromine and bromine c. calcim and oxygen 2. Make a bond angle a. SF CH 4 SF6 CH4 table C. CF₂ C12 d. BiF3. e. Sb₂ CF2 H1₂ вина BiFs Sb2 Answer: K of the rewis dot structure, molecular shape, electron geometry, and Of the following compounds : 08 9. Potassium and chlorine CI 1 0.3 3.0 lewis dot S-6 F-7 molecular shape Octahedral é geometry Octahedral b. promine and bromine Bond angle 90°arrow_forward3. For each molecular fomula, detem ine the electron pair geom etry (EPG), make a 3-dimensional representation, and determ ine the molecular geom etry. Determ ine whether the molecule is polar or non- polar. All of these have central atoms surrounded by terminal atom s/lone pairs only. Formula Lewis structure EPG 3-D drawing Mol Geom. Polar molecule? a. SiCl b. BB13 c. SeF4 d. IF5arrow_forward
- [Molecular shape and polarity] Consider two molecules: XCl3, and YO2 X is an atom in group 5A with electronegativity of 2.0. Y is an atom in group 6A with electronegativity of 2.4. Fill in the blanks. Molecule Lewis structure (Draw an lone pair(s) of electrons, when present) Molecular shape Molecular polarity (Yes: polar or No: nonpolar) XCl3 ? ? ? YO2 ? ? ?arrow_forwardClassify the following bonds as ionic, polar covalent or non-polar covalent 1. C-C bond in H3C-CH3 2. C-H bond in H3C-CH3arrow_forwardCHEMICAL BONDING A. Write the ionic-bond structure of the following compounds. Bal2 Ca3P2 SrSe B. Using the Pauling scale, determine the electronegativity difference to determine the kind of bond present in each compound. Compound Electronegativity Kind of Bond 1st Atom 2nd Atom Difference KI XeF2 BH3 ClO2 NiS C. Draw the Lewis electron dot structure and predict the shape of the following compounds/polyatomic ions. Molecule Total no. of e- Lewis structure Bonding Pairs Lone Pairs Geometry BrF3 PH3 PO3-3arrow_forward
 Introductory Chemistry: A FoundationChemistryISBN:9781285199030Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781285199030Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning





